ACETONE
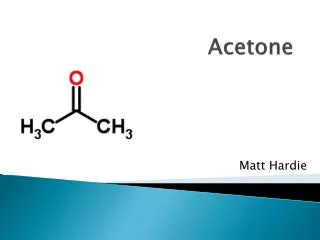
ACETONE. POSTED APRIL 9TH, 2012. BY MAKAYLA HARRIS AND BREANA ZUVER. ABOUT THE MOLECULE OF THE WEEK. CHEMICAL FORMULA: C3H6O OR OC(CH3)2 MOLAR MASS: 58.081 g/mol DENSITY: 791.00 kg/m³ MELTING POINT: -95° C (-139° F) SOLUBILITY IN WATER : Quite Soluble in water. History of Acetone.

ACETONE
E N D
Presentation Transcript
ACETONE POSTED APRIL 9TH, 2012. BY MAKAYLA HARRIS AND BREANA ZUVER
ABOUT THE MOLECULE OF THE WEEK • CHEMICAL FORMULA: C3H6O OR OC(CH3)2 • MOLAR MASS: 58.081 g/mol • DENSITY:791.00 kg/m³ • MELTING POINT: -95° C (-139° F) • SOLUBILITY IN WATER: Quite Soluble in water.
History of Acetone A common building block in organic chemistry, Acetone was first discovered in 1895 by Richard Wolffenstein. A third of the use of Acetone is for a solvent and a quarter of the use is for a precursor to the compound methyl methacrylate. Acetone has been discovered as having extreme flammability and highly unstable characteristics. In addition it easily degraded by UV light, and therefore is a dangerous environmental contaminant.
Health Effects Although acetone has major environmental effects the health risks in humans are low. Acetone is generally recognized as having low acute and chronic toxicity if inhaled and/or ingested. It is commonly found in human urine and blood and varies in levels. In pregnant women and diabetics there is always a higher level of Acetone in their bodies. An increase in Acetone in epileptic victim's diet can result in a decrease of epileptic attacks on the individual.
WORKS CITED • https://www.google.com/search?hl=en&q=acetone%20structure&bav=on.2,or.r_gc.r_pw.,cf.osb&biw=1366&bih=705&um=1&ie=UTF-8&tbm=isch&source=og&sa=N&tab=wi&ei=jfWGT87lJeXSiALU6Zn2Dw • http://portal.acs.org/portal/acs/corg/content • http://en.wikipedia.org/wiki/Acetone