Bioenergetics
Bioenergetics. I. Introduction. A. Definition. B. History. 1. Count Rumford ( 1798 ) work into heat studies , cannon boring. 2. Sadi Carnot ( 1824 ) work depends on the temperature , cyclic engine. 3. James Joule ( 1842 ) established equivalence of work and heat ( Joule ).

Bioenergetics
E N D
Presentation Transcript
Bioenergetics I. Introduction A. Definition B. History
1.Count Rumford(1798) work into heat studies, cannon boring 2.Sadi Carnot(1824) work depends on the temperature, cyclic engine 3.James Joule(1842) established equivalence of work and heat (Joule) 4.Rudolph Classius(1850) states the firsttwo laws of thermodynamics 5.J. Gibbs (1878) proposed free energy or system energy to perform work when To and pressure are uniform throughout a system called Free Energy
6.Ludwig Boltzman late 19th century, proposed energy existed as packets based on particles he called atoms, started fields of thermodynamics and kinetics. Committed suicide in 1906 because no one believed him. 7.Albert Einstein late 1905 atoms held the energy (basis for almost all energy equations) 8.Ernest Rutherford (1910) and Neils Bohr (1912) would design the shape and structure of an atom.
1. Potential Energy = StoredEnergy Figure 8.2
2. KineticEnergy = MotionEnergy Figure 8.2
1. Law ofConservation, Constancy, or Quantityof Energy Figure 8.3
2. Law ofTransformation, Order, or Qualityof Energy Figure 8.3
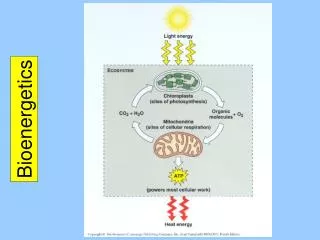
The basis for life on earth is bound to this diagram. Figure 9.2
1. Predicting reaction spontaneity = Free Energy (ΔG) ΔG = free energy ΔH = enthalpy ΔG = ΔH - TΔS ΔS = entropy T =absolute temperature
2. Exergonic reactions = release of free energy 3. Endergonic reactions = absorbs free energy Figure 8.6
1. Structure of ATP Figure 8.8
Cycling of ATP Figure 8.9
2. ATP Coupled Reactions Figure 8.10
II. Enzymes A. Definition B. History
1.Pasteur vs. Leibig (1890’s) Fermentationneeds intact cells or not 2.Buchner late 1890’s Yeast yield EtOH and CO2 from catalyst in cells (yeast) coined the term “enzyme” 3.Sumner (1926) isolated crystalline protein called urease
1. Overview Figure 8.13
2. How? Figure 8.14
2. How? Figure 8.17
3. Kinetics Line Weaver-Burk plot
4. Environmental Effects (Temperature) Figure 8.18
4. Environmental Effects (Inhibitors) Figure 8.19
5. Regulation (Allosteric) Figure 8.20
5. Regulation (Feedback Inhibition) Figure 8.21