Atoms
Atoms. 9.21.2009. What is an Atom?. Just exactly what is an atom and why is it important? An atom is the smallest particle into which an element can be divided and still keep its unique chemical properties Atoms make up elements Atoms are called the building blocks of matter.

Atoms
E N D
Presentation Transcript
Atoms 9.21.2009
What is an Atom? • Just exactly what is an atom and why is it important? • An atom is the smallest particle into which an element can be divided and still keep its unique chemical properties • Atoms make up elements • Atoms are called the building blocks of matter
Just how BIG is an Atom? • Do you think you can see an atom with your eye? • Not at all • An atom is so small that it would take a stack of 50,000 aluminum atoms to equal the thickness of a sheet of aluminum foil
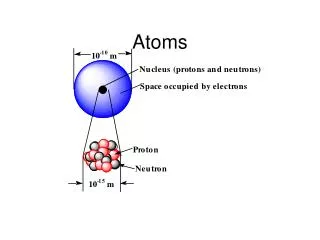
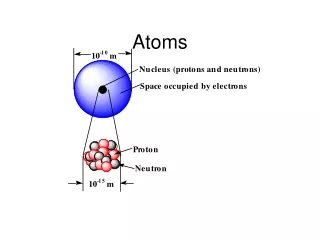
Did you know…. • An atom actually is made up of even smaller particles: • The nucleus • The nucleus is the central part of an atom • It consists of 2 parts: • 1. protons • 2. neutrons • Outside the nucleus • This is where the atom’s electrons are found
The Nucleus • The nucleus is made up of what 2 things? • 1. neutrons • Neutrons are ‘neutral’ • They have NO charge • 2. protons • Protons have a positive charge • Which particles in the diagram represent protons and which represent neutrons?
The Nucleus (Part 2) • The nucleus contains almost all of an atoms mass • Each proton has a mass of 1 atomic mass unit (abbreviated amu) • Each neutron also has a mass of 1 atomic mass unit (amu) • However, the nucleus has a very small volume
Electrons….The Outsiders • Electrons are typically found outside the nucleus within electron clouds • Electrons are high in energy and move around rapidly outside the nucleus • Electrons are negatively charged
It’s Lonely On the Outside • The mass of an electron is very small and considered to be insignificant • In a neutral atom (one with no overall charge) the number of electrons is equal to the number of protons
How Are Electrons Arranged? • Remember that electrons are found outside of an atoms nucleus • They move randomly within regions called electron clouds • Each electron cloud can only hold a certain number of electrons • The first electron cloud only holds 2 electrons • The second and third electron clouds hold 8 electrons each • You have to fill each cloud completely before the electrons can move to the next
The Big Picture • If all atoms are made out of the same things…..then how are they different from one another? • The key is the number of the particles present • The number of protons in an atom determines which element it is • The number of protons is the atomic number of that atom
Atomic Number • All atoms of an element will have the same atomic number • Examples: • Every hydrogen atom has only one proton • Every gold (Au) atom has 79 protons • Look at the periodic table and determine which of the numbers in the box corresponds to the atomic number. • Do you see a pattern?
Atomic Number • You should have noticed that as the elements go from left to right, the atomic number increases by… • 1 • Ifthe atomic number is equal to the number of protons, what does that tell us? • That as we move from left to right, the number of protons in the nucleus also increases by 1
Atomic Number • Using your periodic table, find the element that has: • An atomic number of 41 • An atomic number of 22 • An atomic number of 16 • 31 protons • 18 protons
Atomic Mass • The atomic mass number is the sum of the number of protons plus neutrons in an atom • Why are electrons not included in an atoms atomic mass? • Remember that the mass of an electron is extremely small and insignificant
Atomic Mass • Looking at the picture below and determine which number is the atomic mass
Atomic Mass Examples • Atomic mass = ? • Neutrons + protons • How many neutrons does Yttrium have? • What is the atomic mass of Scandium? • If an atom has 7 protons and 7 neutrons, what is its atomic mass? What element is this?
Atomic Mass Examples • If Hafnium has 72 protons and an atomic mass of 178.5 amu’s, how many neutrons does it have? • Can an atom really have 106.5 neutrons? • NOOO!!!!!!! • The reason it is listed as being a decimal is because of a thing called an isotope
Iso WHO? • Isotopes are atoms that have the same number of protons but have DIFFERENT numbers of neutrons • So when you see a decimal in the atomic mass it is because the atomic mass of each isotope is used to determine an elements average atomic mass • Remember that it is the number of PROTONS that determines the elements