The Nitrogen Cycle
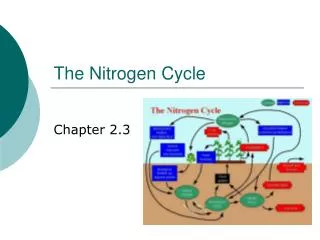
The Nitrogen Cycle. The Nitrogen Cycle. Represents one of the most important nutrient cycles found in terrestrial ecosystems. Model that describes the movement of nitrogen in its many forms between the hydrosphere, lithosphere, atmosphere, and biosphere. The Hydrosphere.

The Nitrogen Cycle
E N D
Presentation Transcript
The Nitrogen Cycle • Represents one of the most important nutrient cycles found in terrestrial ecosystems. Model that describes the movement of nitrogen in its many forms between the hydrosphere, lithosphere, atmosphere, and biosphere.
The Hydrosphere • The hydrosphere describes the waters of the earth. Water exists on the earth in various stores, including the: atmosphere, oceans, lakes, rivers, glaciers, snowfields, and groundwater.
Hydrosphere Continued… • Water moves from one store to another by way of: evaporation, condensation, precipitation, deposition, runoff, infiltration, sublimation, transpiration, and groundwater flow.
continued • The form and movement of nitrogen are greatly influenced by components of the hydrologic cycle, which is particularly important for agriculture and the environment.
The Lithosphere • Rigid outer layer of earth; Includes crust and upper part of mantle. • Relatively strong layer in contrast to underlying asthenosphere.
The brittle most upper layer of the Earth that is broken up into a number of tectonic plates. Consists of the heavy oceanic and lighter continental crusts and the upper part of the mantle. The lithosphere rests on a soft layer called the asthenosphere, over which the plates of the lithosphere glide. Lithosphere Continued…
The Atmosphere • Life on earth is supported by the atmosphere, solar energy, and our planet's magnetic fields. The atmosphere absorbs the energy from the sun, recycles water and other chemicals, and works with the electrical and magnetic forces to provide a moderate climate. The atmosphere • The atmosphere also protects us from high-energy radiation and the frigid vacuum of space.
Composition of Atmosphere • The atmosphere is primarily composed of nitrogen (N2, 78%), oxygen (O2, 21%), and argon (Ar, 1%). • A number of other very influential components are also present: the water (H2O, 0 - 7%), "greenhouse" gases or ozone (O, 0 - 0.01%), carbon dioxide (CO2, 0.01-0.1%).
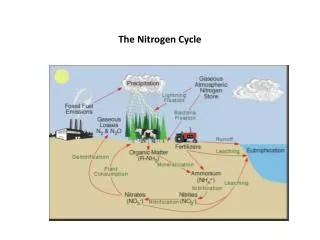
continued • Nitrogen, mostly in the form of ammonium and nitrate, reaches the Earth's surface as a result of atmospheric lightning, precipitation and industrial pollution.
The Biosphere • The biosphere is the life zone of the Earth and includes all living organisms, including man, and all organic matter that has not yet decomposed. • The biosphere is structured into a hierarchy known as the food chain whereby all life is dependent upon the first tier (i.e. mainly the primary producers that are capable of photosynthesis).
Biosphere Part 2 • The biosphere can be divided into distinct ecosystems that represent the interactions between a group of organisms forming a trophic pyramid and the environment or habitat in which they live.
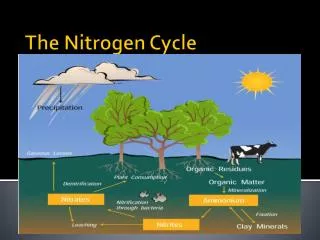
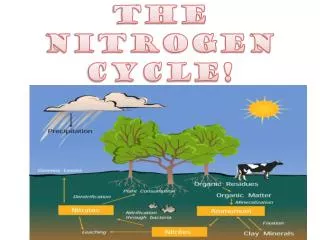
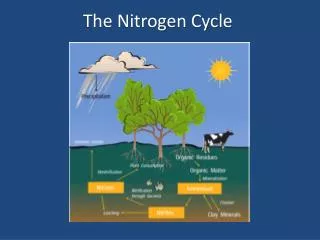
Continued… • Animals consume nitrogen from plants • Plants consume nitrogen from the soil • Soil gets nitrogen from water or rain that contains nitrogen.
Nitrogen Cycle Continued… • All life requires nitrogen-compounds, e.G., Proteins and nucleic acids. • Air, which is 79% nitrogen gas (N2), is the major reservoir of nitrogen. • But most organisms cannot use nitrogen in this form.
Plants must secure their nitrogen in "fixed" form, i.E., Incorporated in compounds such as: Nitrate ions (NO3-) • Ammonia (NH3) • Urea (NH2)2CO • Animals secure their nitrogen (and all other) compounds from plants (or animals that have fed on plants).
Three processes are responsible for most of the nitrogen fixation in the biosphere are … atmospheric fixation by lightning biological fixation by certain microbes - alone or in a symbiotic relationship with plants industrial fixation Nitrogen Fixation
Atmospheric Fixation • The enormous energy of lightning breaks nitrogen molecules and enables their atoms to combine with oxygen in the air forming nitrogen oxides. • These dissolve in rain, forming nitrates, that are carried to the earth. • Atmospheric nitrogen fixation probably contributes some 5-8% of the total nitrogen fixed.
Biological Fixation • The ability to fix nitrogen is found only in certain bacteria. • Some live in a symbiotic relationship with plants of the legume family (e.g., soybeans, alfalfa). • Some establish symbiotic relationships with plants other than legumes (e.g., alders).
Continued… • Some nitrogen-fixing bacteria live free in the soil. • Nitrogen-fixing cyanobacteria are essential to maintaining the fertility of semi-aquatic environments like rice paddies.
Industrial Fixation • Under great pressure, at a temperature of 600°C, and with the use of a catalyst, atmospheric nitrogen and hydrogen (usually derived from natural gas or petroleum) can be combined to form ammonia (NH3). • Ammonia can be used directly as fertilizer, but most of its is further processed to urea and ammonium nitrate (NH4NO3).
Decay • Proteins made by plants enter and pass through food webs just as carbohydrates do. • At each trophic level, their metabolism produces organic nitrogen compounds that return to the environment, chiefly in excretions.
Continued… • The final beneficiaries of these materials are microorganisms of decay. They break down the molecules in excretions and dead organisms into ammonia.
Ammonia can be taken up directly by plants - usually through their roots. Most of the ammonia produced by decay is converted into nitrates. This is accomplished in two steps: Bacteria of the genus Nitrosomonas oxidize NH3 to nitrites(NO2-). Bacteria of the genus Nitrobacter oxidize the nitrites to nitrates (NO3-). Nitrification
Continued… • These two groups or autotrophic bacteria are called nitrifying bacteria. Through their activities (which supply them with all their energy needs), nitrogen is made available to the roots of plants.
Assimilation • Plant roots absorb inorganic ammonia, ammonium ions, and nitrate ions. Formed by nitrification and nitrogen fixation. • Ions are used to make nitrogen containing organic molecules such as: • DNA • Amino Acids • Proteins
Dentrification • The three processes above remove nitrogen from the atmosphere and pass it through ecosystems. • Denitrification reduces nitrates to nitrogen gas, thus replenishing the atmosphere. • Bacteria are the agents. They live deep in soil and in aquatic sediments where conditions are anaerobic. They use nitrates as an alternative to oxygen for the final electron acceptor in their respiration.
Human Influence • German chemist of WWII, Fritz Haber developed a chemical process in which nitrogen and hydrogen gas combine to form gaseous ammonia. • Coupled with irrigation, this input of nitrogen into the soil revolutionized agriculture by increasing crop yields
Ways Humans Intervene… #1 • We emit a large amount of nitrogen into the atmosphere when we burn fuel
… #2 • We emit heat-trapping nitrous oxide gas into the atmosphere through anaerobic bacteria on livestock wastes and commercial inorganic fertilizers applied to the soil • Emission of this gas rise and account for few greenhouse gases that can cause global warming • When it reaches the stratosphere, it depletes some of the ozone layer
…3 • We remove nitrogen from the earth's crust when we mine nitrogen-containing materials for fertilizers • Deplete nitrogen from soil by harvesting nitrogen-rich crops • Leach water-soluble nitrate ions from soil by irrigation
…4 • Remove nitrogen from soil when we burn grasslands and clear forests before planting crops
…5 • Add excess nitrogen compounds to aqautic systems in agricultural runoff, sewage, and deposition of nitrogen compounds from the atmosphere • Stimulates excess growth of algae and other aquatic plants • Breakdown of dead algae by aerobic decomposers deplete water of dissolved oxygen and disrupt aquatic systems and reduce aquatic biodiversity
…6 • Add excess nitrogen compounds to terrestrial ecosystems through atmospheric deposition…
Atmospheric Deposition • The movement of reactive nitrogen compounds, such as nitric acid, nitrogen dioxide, from the atmosphere onto plant leaves and other surfaces
…6 (continued) • The nitrogen becomes available for plant and microbial growth, and can lead to weeds which can better use nitrogen for growth, outgrowing/eliminating other plants that cant use nitrogen as well. • THUS: our excessive inputs of nitrogen into the atmosphere can reduce terrestrial biodiversity
http://liftoff.msfc.nasa.gov/academy/space/atmosphere.html http://www.geog.ouc.bc.ca/physgeog/contents/images/lithosphere.gif http://www.oilandgas.org.uk/issues/images/z0002409.gif http://www.webref.org/geology/1/lithosphere.htm http://www.bartleby.com/65/li/lithosph.html http://www.elmhurst.edu/~chm/onlcourse/chm110/outlines/nitrogencycle.html http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/N/NitrogenCycle.html Works Cited
http://web.geology.ufl.edu/Biosphere.html http://www.cas.muohio.edu/~mbi-ws/biogeochemicalcycles/Nitrogen/nitrogen.htm#Ass http://www.marietta.edu/~biol/102/ecosystem.html#TheNitrogenCycle12 Living in the Environment/Eleventh Edition/G Tyler Miller, Jr. Continued…