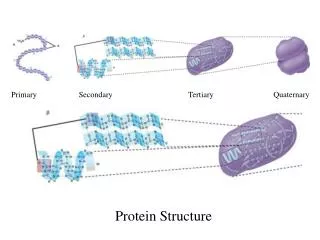
Primary Secondary Tertiary Quatern
150 likes | 486 Views
Primary Secondary Tertiary Quaternary. Protein Structure. Energy. Outline. I. Introduction A. Metabolism B. Energy defined II. Laws of Thermodynamics

Primary Secondary Tertiary Quatern
E N D
Presentation Transcript
Primary Secondary Tertiary Quaternary Protein Structure
Outline I. Introduction A. Metabolism B. Energy defined II. Laws of Thermodynamics III. Energy Transfer in Living Systems A. Chemical Reactions B. ATP
Metabolism • All the chemical reactions that occur in the cell • Capacity to get and use energy to build, store, break down, and eliminate substances necessary for growth and reproduction • Catabolism: reactions that break down large molecules into smaller ones e.g. digestion energy released • Anabolism: reactions that build larger molecules from small ones e.g. protein built from AA subunits energy required
Energy • Definition: the ability to do work, cause change • Energy obtained from some cellular reactions is used for fueling other reactions • Thermodynamics: The study of energy
Laws of Thermodynamics • 1st Law: Energy in the universe is constant • Energy can be transferred or transformed but it cannot be created or destroyed • Also known as the Energy Conservation law • True for a closed system
Heat and Entropy • Heat is energy and can do work (steam engine) • Conversion of any form or energy into heat is not fully reversible, not all heat is usable energy • Heat “lost”=energy no longer capable of doing work • Heat is “low quality” energy • Unusable heat associated with disorder (entropy) • Energy flows from high quality to low quality
Laws of Thermodynamics • 1st Law: Energy in the universe is constant • Energy can be transferred or transformed but it cannot be created or destroyed • Also known as the Energy Conservation law • True for a closed system • 2nd Law: Disorder in the universe is increasing • Entropy: measure of disorder • In a closed system entropy increases
Maintaining Order • Systems tend toward disorder • How do you explain order of a cell, or an organism? • Input of energy required to maintain order
Energy Transfer in Living Systems • Each chemical reaction in a cell has reactants and products • 2 types of chemical reactions: • Exergonic: energy released meaning reactants have more energy than products. Occur spontaneously • Endergonic: energy required meaning reactants have less energy than products • Sometimes these reactions are coupled
Energy Transfer in Living Systems • Metabolic Pathway: series of chemical reactions • Often have reactants which are complexed into one or more intermediates before final products A + B → C → D → E + F Where A, B are reactants C, D are intermediates E,F products Specific enzymes required at each step (each arrow) • Chemical equilibrium: recall this occurs when forward and backward reactions occur at the same rate. Concentrations of reactants and products are stable but not necessarily equal
ATP • Adenosine triphosphate • Nucleotide based molecule- ribose, adenine, and 3 phosphate groups • Cellular energy, powers cellular work • Bonds between phosphate groups can be broken by hydrolysis ATP + H2O ↔ ADP + Pi (inorganic phosphate) + energy • Phosphate groups all negative-repulse each other, like a coiled spring