Ch 12-Metabolism Overview
330 likes | 699 Views
Ch 12-Metabolism Overview. Introduction to Metabolism. A principle task of Biochemistry is to understand how cells regulate its assorted reaction sequences and controls its internal environment Previously, we have looked at this from an individual compound perspective.

Ch 12-Metabolism Overview
E N D
Presentation Transcript
Introduction to Metabolism • A principle task of Biochemistry is to understand how cells regulate its assorted reaction sequences and controls its internal environment • Previously, we have looked at this from an individual compound perspective
Now we switch to reaction sequences, called pathways, the relationship between each pathway and cellular architecture, the biological importance of each pathway, control mechanisms that regulate flux, and experimental methods used to investigate metabolism • Flux- intracellular reaction rates
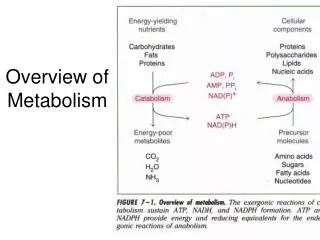
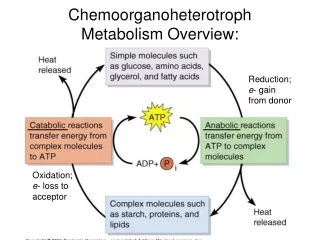
Metabolism Overview Metabolism is divided into two categories: Catabolism- processes related to the degradation of complex substances Anabolism- processes that synthesize complex organic molecules
There are 3 stages associated with both: Stage 1- the interconversion of macromolecules with their monomer parts Stage 2- The interconversion of the monomers with their simpler parts Stage 3- The ultimate degradation to or synthesis from inorganic compounds such as CO2, H2O, NH3
Basic Terms • Intermediary Metabolism- all reactions concerned with storing and generating metabolic energy and with using that energy in biosynthesis of low molecular weight compounds and energy storage compounds • These reactions do not include those that need a nucleic acid template. • The information needed is already embedded in the structure of the enzyme that catalyzes the reaction.
Basic Terms • Energy Metabolism- a part of intermediary metabolism consisting of pathways that store or generate metabolic energy • Central pathways- pathways that are substantially the same in many different organisms, and they account for relatively large amounts of mass transfer and energy generation within a cell.
Synthesis vs. Consume • Most organisms derive both the raw materials and the energy for biosynthesis from organic fuel molecules such as glucose • The central pathways revolve around the oxidation of fuel molecules and the synthesis of small biomolecules from the resulting fragments • This is true for all aerobic organisms
Synthesis vs. Consume • The basic distinction of these organisms lies in the source of their fuel molecules • Autotrophs- synthesize glucose and all other organic compounds from inorganic Carbon, mainly CO2 • Heterotrophs- can synthesize their organic metabolites only from other organic compounds, which they must comsume.
This figure presents metabolism in much more detail than the previous image and is the basic road map for metabolism The middle of the figure, with the larger arrows, are the central pathways of energy metabolism
Glycolysis- a stage 2 pathway for degradation of carbohydrates in both aerobic and anaerobic cells. The major input to glycolysis is glucose. It usually comes from energy storage polysaccharides or dietary carbohydrates Notice that as glucose is converted to pyruvate, a 3 carbon keto acid, an ADP is converted to an ATP!!
Anaerobic organisms further reduce pyruvate to simpler compounds such as ethanol and CO2, through a process called fermentation • In aerobic organisms, the major fate of pyruvate is acetyl-coenzyme A, acetyl-CoA, which is then incorporated into the Citric Acid Cycle (CAC)
The Citric Acid Cycle • The CAC is the principle stage 3 pathway in aerobic organisms. • This cycle accepts simple carbon compounds from carbohydrates, proteins, and lipid degradation and oxidizes them to CO2 • All catabolic pathways converge at the CAC • The oxidation reactions of the CAC generate reduced electron carries (NADH, FADH2) whose reoxidation drives ATP biosynthesis, primarily through processes in the mitochondrial respiration chain.
Other stage 2 pathways also deliver Acetyl-CoA to the CAC • Ex. b-oxidation of fatty acids and the amino acid oxidation pathways • The 2 carbons of Acetyl-CoA don’t have to be oxidized by the CAC • They can also be used as building blocks for anabolic pathways to produce fatty acids, steriods, and amino acids • These and other biosynthesis processes use a reduced electron carrier, NADPH, which is structurally very similar to NADH.
Distinct Pathways for Biosynthesis and Degradation • From the earlier figures, it may appear that most catabolic pathways operate simply as the reversal of the analogous anabolic pathways and/or vice versa. • However, the opposed pathways are quite distinct from one another • They may share common intermediates, but they are separate reactions sequences, regulated by distinct mechanisms, and use different enzymes • Some even occur in different locations!
Example • Fatty acid synthesis takes place in the cytosol while fatty acid degradation through oxidation takes place in the mitochondria. • The biosynthesis and degradation pathways are rarely, if ever, simple reversals of one another!
Reasons for separate pathways • The existence of separate pathways is important for 2 reasons: • Thermodynamics- -Recall that for a pathway to proceed in a given direction, it must be exergonic, in that direction -if it is strongly exergonic, than the reverse direction would be just as strongly endergonic!!
Reasons for separate pathways 2) Control- Using separate pathways for biosynthesis and degradation processes is crucial for control, so conditions that activate one pathway tend to inhibit the opposed pathway. Example: ATP levels Low: -more carbon is oxidized in CAC -Fatty acid and carbohydrate breakdown are activated ATP levels High: -less carbon is oxidized in CAC -Carbon stored as fats and carbs so fatty acid syn. and Gluconeogenesis are activated
Futile Cycle • Futile Cycle- a cycle in which no net work is done. • Separation is important to avoid a futile cycle:
Occasional Exceptions • Occasionally, opposing processes occur in the same location but are controlled by opposing methods. • One example occurs in carbohydrate metabolism. • #1 occurs in glycolysis • #2 occurs in gluconeogenesis • Both occur in the cytosol!!
Substrate Cycle • However, enzymes catalyzing both reactions respond to allosteric effectors, such that one enzyme is inhibited by conditions that activate the other. • This effective control prevents the futile cycle and are referred to as a Substrate Cycle. • Substrate Cycle- two seemingly opposed cellular reactions that are independently controlled.
Substrate Cycles • Substrate cycles represent an efficient regulatory mechanism, because a small change in the activity of either or both enzymes can have a much larger effect on the flux of metabolites.
Bioenergetic Considerations • Recall that thermodynamically unfavorable, endergonic reactions will proceed smoothly in the unfavored direction only if it can be coupled to a thermodynamically favorable, exergonic reaction. • In living systems, most of the energy needed to drive biosynthetic reactions is derived from the oxidation of organic substrates
Oxygen, the ultimate electron acceptor for aerobic organisms, is a strong oxidant; it has a marked tendency to attract electrons and becomes reduced in the process • The energy released is constant whether we burn wood, which is the oxidation of a glucose polymer (cellulose); combust glucose in a calorimeter, or the metabolic oxidation of glucose
The difference is how the energy is released. • When wood is burned, most energy is released as heat with the only useful work being done by a secondary system. • In biological oxidations, the reactions occur without a large increase in temperature and some of the free energy is captured as chemical energy
This chemical energy is captured largely through the synthesis of ATP • The hydrolysis of ATP can then be coupled to many processes to provide this chemical energy for biological work • In the catabolism of glucose, about 40% of the released energy is used to drive ADP + Pi → ATP
Most biological oxidations do not involve direct transfers of electrons from a reduced substrate to oxygen • A series of coupled oxidation-reduction reactions occur with the electrons passed to intermediate electron carriers such as NAD+ and then to oxygen
This reaction sequence is called the electron transport chain, or respiratory chain and oxygen is called the terminal electron acceptor • The potential energy in organic substrates is released in small increments, which makes it easier to control oxidations, more efficient, and easier to capture the energy released.
Make Sure To Read: • Energy Yields, Respiration Quotients, and Reducing Equivalents • ATP as a Free Energy Currency • Thermodynamic Properties of ATP • Differences between ∆G’ and ∆Go’ • Major Metabolic Control Mechanisms
Experimental Analysis of Metabolism • Since metabolism consists of all the chemical reactions, to study it, to subdivide it into something that can be studied, biochemists seek: • 1) to identify reactants, products, and cofactors , plus stoichiometry, for each reaction involved • 2) to understand how the rate of each reaction is controlled in the tissue of origin • 3) to identify the physiological function of each reaction and control mechanism