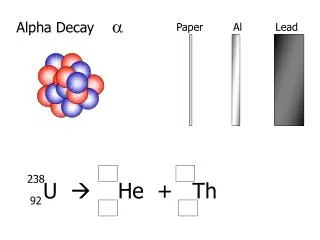
Alpha Decay
Alpha Decay. Readings Nuclear and Radiochemistry: Chapter 3 Modern Nuclear Chemistry : Chapter 7 Energetics of Alpha Decay Theory of Alpha Decay Hindrance Factors Heavy Particle Radioactivity Proton Radioactivity Identified at positively charged particle by Rutherford

Alpha Decay
E N D
Presentation Transcript
Alpha Decay • Readings • Nuclear and Radiochemistry: Chapter 3 • Modern Nuclear Chemistry: Chapter 7 • Energetics of Alpha Decay • Theory of Alpha Decay • Hindrance Factors • Heavy Particle Radioactivity • Proton Radioactivity • Identified at positively charged particle by Rutherford • Helium nucleus (4He2+) based on observed emission bands • Energetics • Alpha decay energies 4-9 MeV • Originally thought to be monoenergetic, fine structure discovered • AZ(A-4)(Z-2) + 4He + Qa
Fine Structure for 228Th decay • Different alpha decay energies for same isotope • Relative intensities vary • Coupled with gamma decay
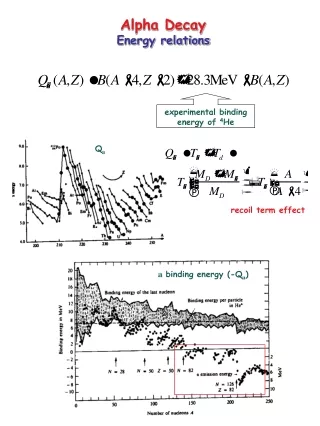
Energetics • Over 350 artificially produced alpha emitting nuclei • Alpha energy variations used to develop decay schemes • All nuclei with mass numbers greater than A of 150 are thermodynamically unstable against alpha emission (Qα is positive) • However alpha emission is dominant decay process only for heaviest nuclei, A≥210 • Energy ranges 1.8 MeV (144Nd) to 11.6 MeV (212mPo) • half-life of 144Nd is 5x1029 times longer then 212mPo Alpha decay observed for negative binding energies
Energetics • Q values generally increase with A • variation due to shell effects can impact trend increase • Peaks at N=126 shell • For isotopes decay energy generally decreases with increasing mass • 82 neutron closed shell in the rare earth region • increase in Qα • α-decay for nuclei with N=84 as it decays to N=82 daughter • short-lived α-emitters near doubly magic 100Sn • 107Te, 108Te, 111Xe • alpha emitters have been identified by proton dripline above A=100
Alpha Decay Energetics • Q value positive for alpha decay • Q value exceeds alpha decay energy • maTa = mdTd • md and Td represent daughter • From semiempirical mass equation • emission of an α-particle lowers Coulomb energy of nucleus • increases stability of heavy nuclei while not affecting the overall binding energy per nucleon • tightly bound α-particle has approximately same binding energy/nucleon as the original nucleus • Emitted particle must have reasonable energy/nucleon • Energetic reason for alpha rather than proton • Energies of alpha particles generally increase with atomic number of parent
Energetics • Calculation of Q value from mass excess • 238U234Th + a + Q • Isotope Δ (MeV) 238U 47.3070 234Th 40.612 4He 2.4249 • Qa=47.3070 – (40.612 + 2.4249) = 4.270 MeV • Q energy divided between the α particle and the heavy recoiling daughter • kinetic energy of the alpha particle will be slightly less than Q value • Conservation of momentum in decay, daughter and alpha are equal rd=r • recoil momentum and the -particle momentum are equal in magnitude and opposite in direction • p2=2mT where m= mass and T=kinetic energy • 238Ualpha decay energy
Energetics • Kinetic energy of the emitted particle is less than Coulomb barrier α-particle and daughter nucleus • Equation specific of alpha • Particles touching • For 238 U decay • Alpha decay energies are small compared to the required energy for the reverse reaction • Alpha particle carries as much energy as possible from Q value, • For even-even nuclei, alpha decay leads to the ground state of the daughter nucleus • as little angular momentum as possible • ground state spins of even-even parents, daughters and alpha particle are l=0
Energetics • Some decays of odd-A heavy nuclei populate low-lying daughter excited states that match spin of parent • Leads to fine structure of alpha decay energy • Orbital angular momentum of α particle can be zero • 83% of alpha decay of 249Cf goes to 9th excited state of 245Cm • lowest lying state with same spin and parity as parent • Long range alpha decay • Decay from excited state of parent nucleus to ground state of daughter • 212mPo • 2.922 MeV above 212Po ground state • Decays to ground state of 208Pb with emission of 11.65 MeV alpha particle • Systematics result from • Coulomb potential • Higher mass accelerates products • larger mass • daughter and alpha particle start further apart • mass parabolas from semiempirical mass equation • cut through the nuclear mass surface at constant A • Explains beta decay in decay chain
Mass parabolas: 235U decay series Beta Decay to Energy minimum, then Alpha decay to different A Branched decay observed (red circles)
Alpha decay theory • Distance of closest approach for scattering of a 4.2 MeV alpha particle is ~62 fm • Distance at which alpha particle stops moving towards the daughter • Repulsion from Coulomb barrier • An alpha particle should not get near the nucleus • Alpha particle should be trapped behind a potential energy barrier Vc Alpha decay energy
Alpha decay theory • Wave functions are only completely confined by potential energy barriers that are infinitely high • With finite size barrier wave function has different behavior • main component inside the barrier • finite piece outside barrier • Tunneling • classically trapped particle has component of wave function outside the potential barrier • Some probability to go through barrier • Closer the energy of the particle to the top of the barrier more likely the particle will penetrate barrier • Increase probability of barrier penetration • Higher alpha decay energy, higher probability to penetrate barrier • Shorter half life with higher alpha decay energy
Alpha Decay Theory • Geiger Nuttall law of alpha decay • Log t1/2=A+B/(Qa)0.5 • constants A and B have a Z dependence. • simple relationship describes the data on α-decay • over 20 orders of magnitude in decay constant or half-life • 1 MeV change in -decay energy results in a change of 105 in the half-life
Expanded Alpha Half Life Calculation • More accurate determination of half life from Hatsukawa, Nakahara and Hoffman • Theoretical description of alpha emission based on calculating the rate in terms of two factors • rate at which an alpha particle appears at the inside wall of the nucleus • probability that the alpha particle tunnels through the barrier • a=P*f • f is frequency factor • P is transmission coefficient Outside of closed shells 78Z82; 100N126 82Z90; 100N126
Alpha Decay Theory • Alternate expression includes an additional factor that describes probability of preformation of alpha particle inside parent nucleus • No clear way to calculate such a factor • empirical estimates have been made • theoretical estimates of the emission rates are higher than observed rates • preformation factor can be estimated for each measured case • uncertainties in the theoretical estimates that contribute to the differences • Frequency for an alpha particle to reach edge of a nucleus • estimated as velocity divided by the distance across the nucleus • twice the radius • lower limit for velocity could be obtained from the kinetic energy of emitted alpha particle • However particle is moving inside a potential energy well and its velocity should be larger and correspond to the well depth plus the external energy • On the order of 1021 s-1
Alpha Decay Calculations • Alpha particle barrier penetration from Gamow • T=e-2G • Determination of decay constant from potential information • Using the square-well potential, integrating and substituting • Z daughter, z alpha
Gamow calculations • From Gamow • Log t1/2=A+B/(Qa)0.5 • Calculated emission rate typically one order of magnitude larger than observed rate • observed half-lives are longer than predicted • Observation suggest probability to find a ‘preformed’ alpha particle on order of 10-1
Alpha Decay Theory • Even-even nuclei undergoing l=0 decay • average preformation factor is ~ 10-2 • neglects effects of angular momentum • Assumes α-particle carries off no orbital angular momentum (ℓ = 0) • If α decay takes place to or from excited state some angular momentum may be carried off by the α-particle • Results in change in the decay constant when compared to calculated
Hindered -Decay • Previous derivation only holds for even-even nuclei • odd-odd, even-odd, and odd-even nuclei have longer half-lives than predicted due to hindrance factors • Assumes the existence of pre-formed -particles • a ground-state transition from a nucleus containing an odd nucleon in highest filled state can take place only if that nucleon becomes part of the -particle and therefore if another nucleon pair is broken • less favorable situation than formation of an -particle from already existing pairs in an even-even nucleus and may give rise to the observed hindrance. • if -particle is assembled from existing pairs in such a nucleus, the product nucleus will be in an excited state, and this may explain the “favored” transitions to excited states • Hindrance from difference between calculation and measured half-life • Hindrance factors between 1 and 3E4 • Determine by ratio of measured alpha decay half life over calculated alpha decay half life
Hindrance Factors • Transition of 241Am (5/2-) to 237Np • states of 237Np (5/2+) ground state and (7/2+) 1st excited state have hindrance factors of about 500 (red circle) • Main transition to 60 keV above ground state is 5/2-, almost unhindered
Hindrance Factors • 5 classes of hindrance factors based on hindrance values • Between 1 and 4, the transition is called a “favored” • emitted alpha particle is assembled from two low lying pairs of nucleons in the parent nucleus, leaving the odd nucleon in its initial orbital • Hindrance factor of 4-10 indicates a mixing or favorable overlap between the initial and final nuclear states involved in the transition • Factors of 10-100 indicate that spin projections of the initial and final states are parallel, but the wave function overlap is not favorable • Factors of 100-1000 indicate transitions with a change in parity but with projections of initial and final states being parallel • Hindrance factors of >1000 indicate that the transition involves a parity change and a spin flip
Heavy Particle Decay • Possible to calculate Q values for the emission of heavier nuclei • Is energetically possible for a large range of heavy nuclei to emit other light nuclei. • Q-values for carbon ion emission by a large range of nuclei • calculated with the smooth liquid drop mass equation without shell corrections • Decay to doubly magic 208Pb from 220Ra for 12C emission • Actually found 14C from 223Ra • large neutron excess favors the emission of neutron-rich light products • emission probability is much smaller than the alpha decay • simple barrier penetration estimate can be attributed to the very small probability to preform 14C residue inside the heavy nucleus
Proton Decay • For proton-rich nuclei, the Q value for proton emission can be positive • Line where Qp is positive, proton drip line • Describes forces holding nuclei together • Similar theory to alpha decay • no preformation factor for the proton • proton energies, even for the heavier nuclei, are low (Ep~1 to 2 MeV) • barriers are large (80 fm) • Long half life
Topic Review • Understand and utilize systematics and energetics involved in alpha decay • Calculate Q values for alpha decay • Relate to alpha energy and fine structure • Correlate Q value and half-life • Models for alpha decay constant • Tunneling and potentials • Hindered of alpha decay • Understand proton and other charged particle emission
Homework Questions • Calculate the alpha decay Q value and Coulomb barrier potential for the following, compare the values • 212Bi, 210Po, 238Pu, 239Pu, 240Am, 241Am • What is the basis for daughter recoil during alpha decay? • What is the relationship between Qa and the alpha decay energy (Ta) • What are some general trends observed in alpha decay? • Compare the calculated and experimental alpha decay half life for the following isotopes • 238Pu, 239Pu, 241Pu, 245Pu • Determine the hindrance values for the odd A Pu isotopes above • What are the hindrance factor trends? • How would one predict the half-life of an alpha decay from experimental data?
Pop Quiz • Calculate the alpha decay energy for 252Cf and 254Cf from the mass excess data below. • Which is expected to have the shorter alpha decay half-life and why? • Calculate the alpha decay half-life for 252Cf and 254Cf from the data below. (use % alpha decay)