Chapter 7 – Moist Air
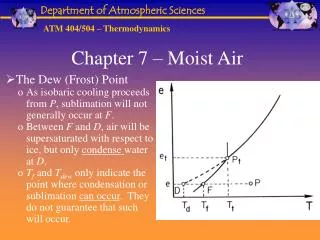
Chapter 7 – Moist Air. The Dew (Frost) Point As isobaric cooling proceeds from P , sublimation will not generally occur at F . Between F and D , air will be supersaturated with respect to ice, but only condense water at D .

Chapter 7 – Moist Air
E N D
Presentation Transcript
Chapter 7 – Moist Air • The Dew (Frost) Point • As isobaric cooling proceeds from P, sublimation will not generally occur at F. • Between F and D, air will be supersaturated with respect to ice, but only condense water at D. • Tf and Tdew only indicate the point where condensation or sublimation can occur. They do not guarantee that such will occur.
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Consider a closed system consisting of dry air, water vapor (moist air), and water (or ice). • The enthalpy of the system can be written as • where we have made use of lv(T) = hv – hw and mt = mv + mw. We can substitute h = cpT for vapor and liquid phase.
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Consider two states of the system linked by an isenthalpic process (H = 0). Each state may be represented by a form of the previous expression. • where md, mt, and const are the same in both states. Then, since H = 0,
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • We can rewrite this as • The denominator on each side is a constant for any (closed) system. Each of the 2 sides of the equation is a function of the state of the system only, i.e., the expression on either side of the equation is an invariant for an isenthalpic process.
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Divide both numerator and denominator in the quotients by md and use the fact that wt = (mv +mw)/md = mt/md to get • Note that (cpd + wtcw) is constant for a given system, but will vary for different systems according to their total water value (liquid + vapor).
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • We can simplify this expression first by neglecting the heat capacity of the water (wt w) yielding, • Now the denominator is no longer constant. Consider wcw as small compared with cpd and make lv a constant, we get
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • In this expression cp can be taken as cpd, or approximated, to a better degree of accuracy, as where the mixing ratio is an average value of w. • Consider the physical process that links 2 specific states (T’, w’) – unsaturated moist air + water – and (T, w) – saturated or unsaturated moist air without water – of the system to which our approximate equation applies. • We have dry air with w gm of water vapor and (w’ – w) gm of liquid water (may or may not be droplets in suspension).
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Assume w’ > w and that saturation is not reached at any time (except eventually when the final state is achieved). • Liquid water evaporates so the mixing ratio increases from w to w’. • As water evaporates, it takes up heat of vaporization from moist air and water (system is adiabatically isolated). • Cooling results reducing temperature from T to T’. • At any instant, the system state differs finitely from saturation – process is spontaneous and irreversible.
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Equivalent temperature (isobaric equivalent temperature) Tei, is defined as the temperature moist air would reach if it were completely dried by condensation of all its water vapor. • Water is withdrawn in a continuous fashion, process is performed isobarically, and system is thermally isolated. • The formulation assumes no liquid water initially and starts from an infinitesimal variation dH, condensing
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • In this expression, dmv is considered negative, i.e., we are condensing a mass dmv of vapor to liquid. • Having condensed this infinitesimal amount of liquid, we remove it before condensing any more vapor. • The enthalpy of the system will decrease by hwdmv, but T is not affected. • For the next infinitesimal condensation, the equation is valid with a new value of mv. • Our equation describes the process with mv as a variable.
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Dividing through by md, and rewriting the differentials as differences, we get • Assuming the latent heat to be constant, we get • Assuming Ti = T, Tf= Tei, wi = w, and wf = 0, we get • Leading to
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Next we consider the Wet-Bulb Temperature, defined by the following process, the reverse of Tei. • We are following a process that goes from any mixing ratio, w, to saturation, wsw. • Closed, adiabatic system with unsaturated moist air and liquid water, temperature T, and total mixing ratio, wt • Water will evaporate drawing heat from moist air • Change in T related to change in wt due to evaporation • When saturation is reached, the temperature is the isobaric wet-bulb temperature, Tw.
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Examples: • A sling (wet-bulb) psychrometer used to calculate RH • Evaporative cooling of air by rain falling through unsaturated region below cloud base • Have the same equation to start with, same assumptions. • Here we have wt instead of w because we have liquid water. • Assuming md >> mt (= mv + mw), Tf = Tw (the limiting temperature), Ti = T, wi = w, wf = wsw, and wtcw << cpd, we get
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • Note that Tei can be approximated (assuming wcw < cpd) as • We see that there is a relation between equivalent and wet-bulb temperatures, to wit • The definition of wet-bulb temperature also lends itself to the defining of a relationship between Tw and Tdew using the approximate form for w e/p and cp = cpd + wtcw,
Chapter 7 – Moist Air • Equivalent and Wet-Bulb Temperatures • In general the relationships between e and T can be given by • The adiabatic isobaric process we are discussing occurs along a straight line with slope . • Extending the line to increasing T will intersect at Tei, where e = 0.
Chapter 7 – Moist Air • Moist Air Adiabats • Poisson’s equation for adiabatic ascent (descent) between (p, T) and (p’, T’) is • Noting that m = d(1 – 0.26q) varies with the moisture content, the adiabats passing through a point p, T will be different for different values of q (w). • Since m d, the adiabats for moist air will be slightly less steep than those for dry air (T will vary slightly more slowly). HOWEVER:
Chapter 7 – Moist Air • Moist Air Adiabats • The potential temperature for moist, unsaturated air is • If we substitute T = (1000/p)-d for T we get • If p = 100 mb and q = 0.01 kg/kg, we get m = 0.9983. • If p = 900 mb and q = 0.01 kg/kg, we get m = 0.9999. • This tells us we can treat moist ascent as dry ascent. • Substitute Tv for T we have the virtual potential temp, v.
Chapter 7 – Moist Air • Moist Air Adiabats • This also leads to a statement of the moist (unsaturated) adiabatic lapse rate. • We have to account for the moisture in the value of the specific heat capacity of moist air, • We apply the binomial theorem to the denominator to give • As with the potential temperature, this says that moist air cooling adiabatically, cools less rapidly than dry air.
Chapter 7 – Moist Air • Saturation of Air by Adiabatic Ascent • The equation for the saturation temperature at the LCL is, • On the diagram, P is the initial state, arrow indicates adiabatic ascent starting from P. • As T changes, esw changes according to arrow from S along saturation curve. • Two arrows will meet at Ts (not shown). • Ts is solved for iteratively.
Chapter 7 – Moist Air • Reversible Saturated Adiabatic Ascent • The system under consideration is a parcel of cloud that rises and expands adiabatically and reversibly, indicating that all condensed water is kept (a closed system). • Since it is adiabatic and reversible, the process is isentropic. • What we derive here is valid for an ice cloud as well. • The expression for the entropy of this system is
Chapter 7 – Moist Air • Reversible Saturated Adiabatic Ascent • If we divide by md and note that the entropy S = const, • Where wt,w = mt/md = wsw + mw/md and the subscript w indicates saturation values (except in mw). • If we differentiate the expression, we get • This equation describes the saturated adiabatic process. • If T and pd are independent variables, wsw = esw/pd and esw = f (T), so the equation determines a curve in T, pd plane.
Chapter 7 – Moist Air • Reversible Saturated Adiabatic Ascent • We can make several assumptions to get an approximate and somewhat simpler form of the equation • Assume wt,w < cpd and esw < pd. This yields • Next assume that lv varies slowly with T, we differentiate • Use of the approximate form is good to within a few percent.
Chapter 7 – Moist Air • Pseudoadiabatic Ascent • In saturated adiabatic ascent, the equations depend on wt. • While the saturation vapor mixing ratio, wsw, is determined, the liquid water mixing ratio, mw/md is arbitrary. • This creates the situation where on a T, p diagram where saturation is just reached, there will be an infinite number of reversible saturated adiabats passing through, each differing ever so slightly from each other, depending on the amount of liquid water in the parcel. • This dilemma can be avoided with a simple assumption.
Chapter 7 – Moist Air • Pseudoadiabatic Ascent • We assume that all the condensed liquid water (or ice) falls out of the parcel as soon as it appears. • This is a process of saturated expansion, but the system is now open and irreversible. • It is called a pseudoadiabatic process and gives unique curves through each T, p point on a diagram. • Assume that mw = 0 and wt,w = ww, yielding • The cooling is slightly greater in pseudoadiabatic than in reversible expansion for the same change in pressure.
Chapter 7 – Moist Air • Equivalent Potential Temperature • Go back to our isentropic equation • Keep latent heat as lv = lv(T) and differentiate • We can introduce a potential temperature of the form • Logarithmically differentiating this we get
Chapter 7 – Moist Air • Equivalent Potential Temperature • Comparing this with our other differential we see that • If we integrate this we have • We can get rid of the constant by taking the value of ’ to be equal to e when all vapor has condensed , wsw = 0
Chapter 7 – Moist Air • Equivalent Potential Temperature • What we have is the representation of a reversible saturated adiabatic process where all the water vapor has condensed out (take parcel to a very cold temperature). • e is the equivalent potential temperature (conserved). • As the equation stands, it applies to saturated air. • We can apply it to unsaturated moist air by lifting the parcel to the LCL, yielding
Chapter 7 – Moist Air • Summary of Temperatures
Chapter 7 – Moist Air • Summary of Temperatures