Elephant Toothpaste
Elephant Toothpaste. By: Ashley Morgan. Purpose. The purpose of this lab was to mix two solutions together and form a foam fountain. When performing this experiment, just by changing one material can give you similar results. Funnel.

Elephant Toothpaste
E N D
Presentation Transcript
Elephant Toothpaste By: Ashley Morgan
Purpose • The purpose of this lab was to mix two solutions together and form a foam fountain. • When performing this experiment, just by changing one material can give you similar results.
Funnel • A tube or pipe that is wide at the top and narrow at the bottom. • It is used for guiding a liquid into a small opening.
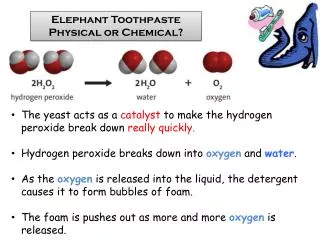
Catalyst • A catalyst is a substance that increases the rate of a chemical reaction without undergoing any permanent chemical change. • In simpler words: • Helps a reaction happen faster • Yeast • Potassium Iodide
Exothermic • Releases heat and causes the termperature of the immeidate surroundings to rise.
Procedure #1 • In a 16 oz bottle mix • 8 drops of food coloring • 1 tablespoon of dish soap (dawn) • ½ a cup of 3% hydrogen peroxide • 3 tablespoons of water • 1 packet of dry yeast (red star) • Watch reaction
Observations • Hydrogen peroxide molecules are very unstable and naturally decompose into water & oxygen gas.
Observations • The yeast acted as a catalyst to remove the oxygen from the hydrogen peroxide. • Since it did this very fast, it created lots of bubbles! • This solution had a scent of freshly baked homemade bread. • Each tiny foam bubble is filled with oxygen.
Observations • The food coloring changed the foam to a red-yellow color. • The experiment created a reaction called an exothermic reaction. • That means it not only created foam but also heat as well. • It also created bubbles, which is a gas.
Question #1 • Why is warm water best to use in this experiment? • Warm water is best for yeast because yeast is a living organism that likes to live in similar temperatures to humants • Put the yeast in hot water and it dies • Put the yeast in cold water and it will be too cold to reproduce.
What is actually happening? • Hydrogen peroide is water with one extra oxygen. • When yeast is added, it acts as a catalyst to split the hydrogen peroxide into water & oxygen. • Bubbles of oxygen are trapped in the soap, which transforms into the foam fountain. • The product formed is just soap, water, and oxygen.
Procedure #2 • Mix in a 16 oz bottle • 40 ml of dish soap (dawn) • 80ml of 3% hydrogen peroxide • 1 tablespoon of potassium iodide • 8 drops of food coloring • 3 tablespoons of warm water • Watch reaction
Observations • The overall equation for this reaction is: • The dishwashing detergent captures the oxygen as bubbles. • Food coloring changed the foam to a blue-green color.
Observations • This had a soapy scent of apples due to the dishwashing soap. • The experiment created an exothermic reaction as well. • The bottle was warm as well. • Bubbles were produced.
Similarities Differences • The both started producing foam right away once the hydrogen peroxide was added. • The bottles was warm. • Bubbles were produced. • Scents • Apples • Bread • Potassium Iodide experiment looked watery rather than bubbly
Conclusion • This experiment used hydrogen peroxide and potassium idodie/yeast mixed with dish soap and water. • If you add all these together, the hydrogen peroxide’s decomposion is sped up the catalyst. • Oxygen is given off and forms foam with the dish soap. • The foam pushes up in the bottle and comes out the top looking like toothpaste.
Works Cited • http://www.using-hydrogen-peroxide.com/elephant-toothpaste.html • http://chemistry.about.com/od/chemistrydemonstrations/a/elephanttooth.htm • http://www.sciencebob.com/experiments/toothpaste.php • http://www.wikihow.com/Make-Elephant-Toothpaste