Energetics
E N D
Presentation Transcript
Energetics Energy changes in chemistry.
Enthalpy • What is enthalpy? • Enthalpy (H) is the total energy of system. It is in the molecules, the bonds, and stored chemically. • Enthalpy change (Δ H) is the measure of the change in the potential energy of the bonds.
Endothermic vs exothermic • What is the difference between the two? • Endothermic reactions have more energy stored in the new bonds (products) than were in the old bonds (reactants) which takes in energy. • Exothermic reactions have more energy in the old bonds (reactants) than in the new bonds (products).
Enthalpy change • What is the value of enthalpy change for exothermic reactions? • Exothermic reaction enthalpy change is negative due to Δ H = enthalpy products – enthalpy reactants. • Endothermic reaction would be the opposite because of the same equation.
Enthalpy change • How do we show enthalpy change of a reaction? • It is usually written with the equation. • + represents endothermic, - represents exothermic • It is given in the units of kJ/mol (or just kJ) because it changes with the amount of reactants and limiting reactants. • This is at thermochemicalstandard conditions which are 25 degrees C, 1 atm and solution concentrations of 1 mol/dm3.
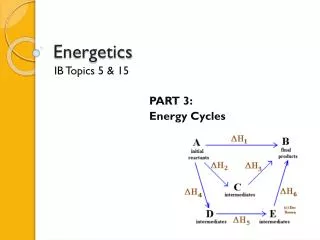
Hess’s Law • What does Hess’s Law say? • It states that if you can add 2 or more different equations to produce the final equation, you can add the individual enthalpy changes to find the total. • This means that if you break down the steps involved, the overall change will be the same.
Hess’s Law • 2H2O2(l) 2H2O(l) + O2(g) • 2H2(g) + O2(g) 2 H2O(l)ΔH= -572 kJ • H2(g) + O2(g) H2O2(l)ΔH= -188 kJ • Where do we start?