Energetics
Energetics. 5.1 Endothermic and Exothermic Reactions. Endothermic rxn heat is taken in from the surroundings ( rxn vessel gets cooler) Exothermic rxn rxn that result in the release of heat ( rxn vessel gets warmer)

Energetics
E N D
Presentation Transcript
5.1 Endothermic and Exothermic Reactions • Endothermicrxn heat is taken in from the surroundings (rxn vessel gets cooler) • Exothermic rxn rxn that result in the release of heat (rxn vessel gets warmer) • Enthalpy change amount of heat energy taken in/ given out in a chemical rxn (ΔH) • Positive for endo. Rxn • Negative for exo. rxn
Stability • Exo. produces a more stable product • Ex. Cdiamond CgraphiteΔH= -1.9 kJ mol-1 • Kinetic vs. Thermodynamic stability • Graphite is more thermodynamically more stable than diamond • Diamond is kinetically stable • What does this mean?
Activation Energy Define Activation Energy
Activation energy • High AE= faster or slower rxn? • Can the conditions be altered? • If yes, in what ways? • Does how endo- or exothermic a rxn is tell us how quickly the rxn will run? • Ex. Which rxn will run faster? • -52 kJ mol-1 • -16 kJ mol-1 • Do endo. or exo. rxns usually occur spontaneously? (under normal conditions)
5.2 Calculations of enthalpy changes from experimental data • Specific heat capacity (c) energy required to raise the temp. of 1 g of substance by 1 K (1°C) or, the energy to raise 1 kg of substance by 1 K. • Units-> J g-1 K-1 J g-1 °C-1 kJ kg-1 K-1 J kg-1 K-1
Calculating c • q= mcΔT • q= heat energy • m= mass • ΔT= change in temperature • How does heat capacity affect how easily a substance can be heated? • Can c be calculated for a substance undergoing cooling?
Measuring enthalpy change of combustion rxns • Worked ex. Page 185 • Literature value for ΔH ethanol is -1371 kJ mol-1 • What are some reasons the calculated value is different? • A bomb calorimeter could have been used so that the system was heavily insulated and provide a plentiful supply of oxygen
Enthalpy changes in solution • General method for measuring • Measure known amounts of reagents • Record initial temps • Mix in a polystyrene cup • Record max/ min temperatures observed • Assume that c for the final solution is the same as water
ΔH of Solutions:Definitions • Enthalpy change of neutralisation (ΔHn) enthalpy change when 1 mol of water molecules are formed when acid reacts with alkali under standard conditions • H+(aq) + OH-(aq) H2O(l) • Enthalpy change of solution (ΔHsol) the enthalpy change when 1 mol of solute is dissolved in excess solvent to form a solution of ‘infinite dilution’ under standard conditions • NH4NO3(s) NH4+(aq) + NO3-(aq)
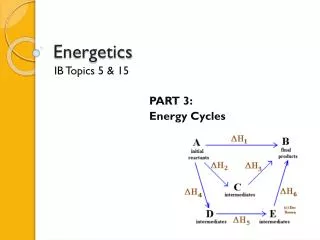
5.3 Hess’s Law • The enthalpy change accompanying a chemical reaction is independent of the pathway between the initial and final states • What does this mean? • Ex. Find ΔHr for the reaction of AB • Knowns: • ΔHr= ΔH1 +ΔH2 • AC =ΔH1 • BC= ΔH2 • What is C? • What needs to change about the BC step?
Hess’s law:Definitions • State function pathway does not matter • Standard conditions pressure= 1 atm (or, 1.01E5 Pa), 298K (or, 25°C) • Standard enthalpy change (ΔHrΘ) the enthalpy change when molar amounts of reactants as shown in the stoichiometric equation react together under standard conditions to give products (Θ= under standard conditions)
Working out enthalpy changes • Hess’s law can be used to determine enthalpy changes of unknows from knowns • P. 194 Worked example
5.4 Bond enthalpies • The enthalpy change when 1 mole of covalent bonds, in a gaseous molecule, are broken under standard conditions (aka bond energy) • Ex. Then enthalpy of H-H bond is 436 kJ mol-1 H2(g) 2H(g) ΔHΘ= +436 kJ mol-1 How many H-H bonds were broken?
Bond Enthalpy • What state must a substance be in to calculate bond enthalpy? • Consider this process: Br2(l) 2Br(g) Br-Br= 193 kJ mol-1 What is the ΔHΘ? +224 kJ mol-1 Why is this higher than the bond enthalpy? The reactants are not in a gaseous state We must also account for the energy required for vaporisation of the reactants This process is called atomisation
Bond breaking • Bond breaking is… endothermic or exothermic? Endothermic! What does that mean about ΔH? Positive! What will bond making be? Exothermic with a negative ΔH
Average bond enthalpy • The average amount of energy required to break 1 mole of covalent bonds, in a gaseous molecule under standard conditions • These are the values used to calculate bond enthalpies
Using bond enthalpies to work out enthalpy changes in a rxn • Must draw out the structural formulas for rxn • Imagine the rxn happening and ALL bonds being broken • Add up the total energy for all broken bonds • Draw in all the bonds formed in products • Add up the total energy of all bonds made • Determine signs for the total enthalpy changes • Broken positive • Made negative • Add the changes to get the overall enthalpy change of the rxn
Example • Consider the rxnbetween ethene and bromine, to produce 1,2-dibromoethane, C2H4(g) + Br2(g) C2H4Br2(g) What bonds are broken? What bonds are made? Follow your steps!
Using a cycle • Same concept as previous calculations, but a process is drawn out to see all the steps • P. 203
5.5 Calculating enthalpy changes:Definitions • Standard enthalpy change of combustion (ΔHcΘ) the enthalpy change when 1 mole of a substance is completely burnt in oxygen under standard conditions. • If ΔHcΘis always negative, what does this mean? • Standard enthalpy change of formation (ΔHfΘ) the enthalpy change when 1 mole of the substance is formed from its elements in their standard states under standard conditions • Endo and exorxns are dependent on the type of substance • ΔHfΘ for any element in its standard state is zero
Using ΔHcΘto calculate enthalpy change • Method 1: Construct an enthalpy cycle • P. 208 • Method 2: rearrange the equations to give the overall equations related to the enthalpy change • P. 209 • Method 3: use an enthalpy level diagram for calculations • P. 210 • Method 4: use the equation, ΔHr = ΣΔHc (reactants)- ΣΔHc(products)
Using ΔHfΘto calculate other enthalpy chages • Method 5: similar to method 1, but used for formation rather than combustion • P. 214 • Method 6: refer method 2 (be sure equations are running in the correct direction) • Method 7: draw enthalpy level diagram for formation (method 3) • Method 8: use the equation, ΔHr = ΣΔHf (products)- ΣΔHf (reactants)
Choosing your method • Choose a method based on the data you are given, NOT on what needs to be found • If needing the enthalpy of combustion and given the enthalpy of formation, use one of the methods 5-8 • Once the basic principle of the methods are understood, there is no need to have any distinctions between them
5.6 Enthalpy changes for ionic compounds • First ionisation energy • Second ionisation energy • First electron affinity enthalpy change when one electron is added to each atom in 1 mol of gaseous atoms under standard conditions (always EXOTHERMIC) X (g) +e- X- (g) • Second electron affinity (always ENDOTHERMIC) why? • Lattice enthalpy(ΔHΘlatt) the enthalpy change when 1 mol of an ionic compound is broken apart into iest constituent gaseous ions under standard conditions
Born-Haber cycles • Enthalpy level diagram breaking down the formation of an ionic compound into a series of simpler steps • 1. put the equation for the enthalpy of formation • 2. add lattice enthalpy • 3. convert to gaseous form (why?) • Two steps • Must convert ALL reactants to gaseous form • Connect the cycle by adding the electrons removed from one reactant to the more electronegative reactant
Draw a Born-Haber cycle • P. 219-221 • Na and Cl example
Comparisons of lattice enthalpy • P. 224 • What is lattice enthalpy the result of? Electrostatic attractions of + and – ions • If the attractions of great, will more or less energy need to be supplied to break the bonds? More
Effect of charge and size • How does the charge of the ions effect lattice enthalpy? The higher the ion charge, the greater the lattice enthalpy • Does NaCl or MgCl2 have great lattice enthalpy? MgCl2 • How does size effect lattice enthalpy? The larger the ions the weaker the forces, the smaller the lattice enthalpy • Which has the larger lattice enthalpy, CsCl or NaCl? NaCl
Theoretical vs. experimental • Theoretical assumes a totally ionic model • What is this? Bonding is solely due to attractive forces between oppositely charged ions • Experimental use the Born-Haber cycle to find • These are compared to determine how ionic a particular compound is
How to use theoretical and experimental values • If values are exactly the same, complete ionic bonding is suggested • If values are significantly different, it is suggested that the bonding has a significant degree of covalency • Ex. Silver iodide Theoretical value/ kJ mol-1 736 Experimental value/ kJ mol-1 876 What do the values suggest?
Covalent character • What is covalent character the result of? Polarisation of the negative ion by the positive one • How does size of the anion effect this? The polarisation effect is greater
5.7 ENtropY • A measure of randomness or disorder of a system • Especially significant in the case of endothermic processes occurring at standard conditions (ice melting at room temp, water evaporating, NaCl dissolving in water etc) • Endo rxns can only occur if there is an increase in entropy • Represented by S • Units: J K-1 mol-1
Standard entropy • Represented by SΘ • Positive ΔSΘ indicates increased entropy • Less order • Negative ΔSΘ indicates decreased entropy • More order
Predicting sign of entropy change • Which state of matter has the higher entropy? Gases • Which state has the least entropy? Solids
Predicting sign • Must consider whether the system’s disorder increases or decreases • Good to consider whether moles of gas have increased or decreased • What would an increase in moles of gas mean? • If moles of gas remain constant, our prediction of a change in entropy would be approximately zero
Calculating entropy change for a rxn ΔSΘ= ΣSΘproducts -- ΣSΘreactants
5.8 Spontaneity • Spontaneous reaction one that occurs without any outside influence
Predicting spontaneity • A reaction being spontaneous does not mean it will run quickly! • Whether a rxn is spontaneous or not under a certain set of conditions can be deduced by looking at the change in the “entropy of the Universe” ΔSUniverse = ΔSsuroundings + Δssystem If ΔSUniverseis positive, the entropy of the universe increases and the rxn occurs spontaneously When heat is given out in a rxn, the entropy of the surroundings get hotter
Gibbs free energy • Represented by ΔG • Also called free energy change • ΔG = ΔH – TΔS • ΔH and ΔS are referring to the system • Under standard conditions this symbol is used: ΔGΘ • For a reaction to be spontaneous, ΔG for the rxn must be NEGATIVE • Units kJ mol-1
Calculating ΔG • Method 1 use ΔGΘ = ΔHΘ – TΔSΘ • Method 2 use ΔGΘ = ΣΔGfΘ(products) – ΣΔGfΘ(reactant) • Standard free energy of formation the free energy change for the formation of 1 mol of substance from its elements in their standard state under standard conditions • Temp must be in K • If no temp is given and it is standard conditions, assume 298K
Non-spontaneous rxns • If a rxn is non-spontaneous, does that mean it will never happen? NO • What will make it run? Outside influence such as; temp, catalysts, etc.
Effects of temp on spontaneity • Refer to the equation: • ΔG = ΔH – TΔS • If ΔS is positive, temp must be high or low to be spontaneous? • High • ΔG must be negative to be spontaneous, thus TΔS must be higher than ΔH
Temp and spontaneity • If ΔS is negative --TΔS will be positive and the rxn cannot be spontaneous • Endo rxns will only occur spontaneously in entropy is increased and temp is significantly high • Exorxns will always be spontaneous at some temp • If the rxn involves a decrease in entropy the rxn will be spontaneous at a lower temp; becomes less spontaneous as temp increases