Kinetic Theory
Kinetic Theory. 16.1. Warm up. Name the states of matter that you know. Give an example from your daily life about the states of matter that you mentioned. Objectives. Explain the kinetic theory of matter. Describe particle movement in the four states of matter.

Kinetic Theory
E N D
Presentation Transcript
Kinetic Theory 16.1
Warm up • Name the states of matter that you know. • Give an example from your daily life about the states of matter that you mentioned.
Objectives • Explain the kinetic theory of matter. • Describe particle movement in the four states of matter. • Explain particle behavior at the melting and boiling points.
Introduction • What happens to molecules of water as ice melts? • Why does snow melts when you hold it in your hand? • What happen when water boils?
Kinetic Theory • Look at the following figure. Can you identify the states of water present? How are these states alike and different?
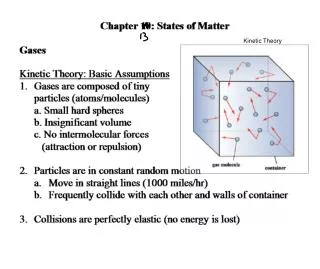
Kinetic Theory • Kinetic theory is an explanation of how particles in matter behave. • To explain the behavior of particles, it is necessary to make some basic assumptions.
Thermal energy • How can frozen solid ice cube have motion? Remember to focus on the particles. • Thermal energy is the total energy of material’s particles, including kinetic (movement) and potential (resulting from forces that act within or between particles)
Average kinetic energy • What is your concept about temperature? • In science, temperature means the average kinetic energy of particles in a substance, or how fast the particles are moving. • Molecules have kinetic energy at all temperatures even at absolute zero or -273.15 oC. • How are kinetic energy and temperature related?
Solid state • Describe the particles in a solid. • Most solid materials have specific type of geometric in which they form when cooled. • Chemical and physical properties of solids often can be attributed to the type of geometric arrangement that the solid forms.
How can a solid changes into a liquid? • What is a melting point? • What is heat of fusion?
Liquid state • Describe the particles in a liquid. • What is a melting point? • What is heat of fusion.
Evaporation • What is evaporation?
Boiling • What is boiling?
Boiling and heat of vaporization. How does external pressure affect the boiling point of a liquid?
Gases • Describe the molecules of a gas. • What happens when you put 5 ml gas in a 20 ml container? How? • What is diffusion?
But what happens if you raise the temperature to super-high levels…between 1000°C and 1,000,000,000°C ? Will everything just be a gas?
Plasma • A plasma is an ionized gas. • A plasma is a very good conductor of electricity and is affected by magnetic fields. • Plasmas, like gases have an indefinite shape and an indefinite volume. • Plasma is the • common state • of matter
Some places where plasmas are found… 1.Flames
Amorphous solids • Ice melts at:0oC • Gold melts at 1064 oC • Lead melts at 327 oC • Some solids have no specific melting point, they soften and directly become liquid. Those solids are called amorphous solids. • Amorphous solids don’t have ordered arrangement of particles. • Examples of amorphous solids are glass and plastic rocks obsidian (volcanic glass)
Liquid crystals • Are another group of materials that do not change state in the usual manner. • Have geometric order in the solid state • liquid crystals are highly responsive to temperature and electric fields. • Example are the displays of watches , clocks, calculators, some notebook computers…
Wrap up • Explain the kinetic theory of matter • Define thermal energy • How are kinetic energy and temperature related? • Describe the particle movement, shape, and volume in the four states of matter • Compare between melting and boiling points • Distinguish between heat of fusion and heat of vaporization • Compare between evaporation and boiling • Compare between state changes of water using the temperature Vs. heat graph • Explain why diffusion in gases is faster than in liquids/ solids • Explain why amorphous solids melts over a temperature range • List examples of amorphous solids.
Assessment • H.W page: 483 # 1 till 7 • SWQ 1: 1- 15.1: Composition of Matter 2- 15.2: Properties of Matter 3- 16.1: Kinetic Theory