Understanding Chemical Compounds: Atoms, Subscripts, and Molecules
This guide explores the structure of chemical compounds made of different atoms, focusing on the role of subscripts and coefficients in molecular formulas. For instance, H2O represents water, while C6H12O6 denotes glucose. Subscripts indicate the number of each type of atom in a molecule, such as in MgCl2 and Al(NO3)3. Coefficients show the number of molecules present, as exemplified by 5NaCl. This resource provides clarity on counting atoms and understanding polyatomic ions, along with practice questions to reinforce learning.

Understanding Chemical Compounds: Atoms, Subscripts, and Molecules
E N D
Presentation Transcript
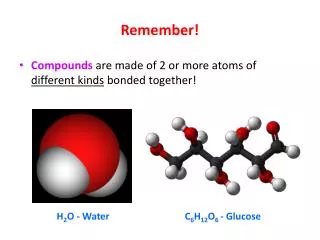
Remember! • Compounds are made of 2 or more atoms of different kinds bonded together! H2O - Water C6H12O6 - Glucose
Biochemistry Counting Atoms
Subscripts • Written below the level of the line • Signify the number of atoms in the molecule 1 Mg atom + 2 Cl atoms MgCl2 • Polyatomic Ions: group together as one • Written in parenthesis • You must multiply through!
Subscripts • Al(NO3)3 There is 1 Al, 3 N, & 9 O! • (CH3)3CH There are 4 C & 10 H!
Coefficients • Written to the left of the molecule • Signify the number of molecules 5NaCl 5 molecules of NaCl 10 total atoms • 3NO2 contains 3 N & 6 O! C6H12O6 – Glucose 2C6H12O6 – 2 Glucose molecules!
Counting Atoms • Practice Qs