SPECTROSCOPY
SPECTROSCOPY . Spectral Distribution of Radiant Energy Wave Number (cycles/cm). SPECTROSCOPY. V = Wave Number (cm-1) l = Wave Length C = Velocity of Radiation (constant) = 3 x 10 10 cm/sec. u = Frequency of Radiation (cycles/sec) The energy of photon:

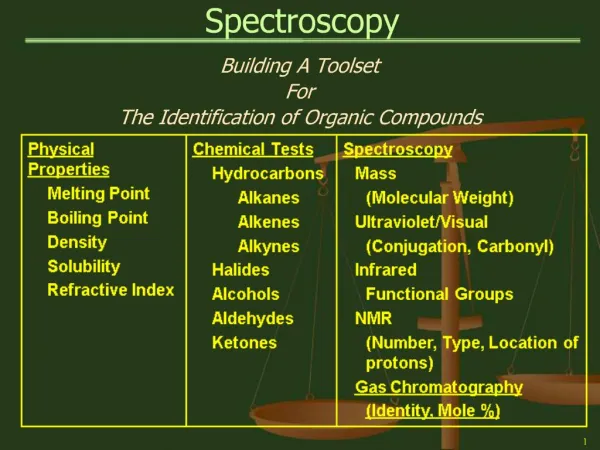
SPECTROSCOPY
E N D
Presentation Transcript
SPECTROSCOPY Spectral Distribution of Radiant Energy Wave Number (cycles/cm)
SPECTROSCOPY V = Wave Number (cm-1) l = Wave Length C = Velocity of Radiation (constant) = 3 x 1010 cm/sec. u = Frequency of Radiation (cycles/sec) The energy of photon: h (Planck's constant) = 6.62 x 10-27 (Ergsec) C = u
DISPERSION OF POLYCHROMATIC LIGHT WITH A PRISM Prism - spray out the spectrum and choose the certain wavelength (l) that you want by slit.
SPECTROSCOPY 1. Spectrophotometer - an instrument which can measure the optical density of a sample at any wavelength.
Fluorometer 2. Fluorometer - measures the intensity of fluorescent light emitted by a sample exposed to UV light under specific conditions.
BEER LAMBERT LAW As the cell thickness increases, the intensity of I (transmitted intensity of light ) decreases.
R- Transmittance R = I0 - original light intensity I- transmitted light intensity % Transmittance = 100 x Absorbance (A) or optical density (OD) = Log = Log = 2 - Log%T Log is proportional to C (concentration of solution) and is also proportional to L (length of light path through the solution). I0 1 I T I I I I0 I0 I0
A CL = KCL by definition and it is called the Beer Lambert Law. A = KCL K = Specific Extinction Coefficient ---- 1 g of solute per liter of solution A = ECL E = Molar Extinction Coefficient ---- Extinction Coefficient of a solution containing 1g molecule of solute per 1 liter of solution
E differs from K (Specific extinction Coefficient) by a factor of molecular weight. UNITS A = ECL A = No unit (numerical number only)
Liter K= Cm Gram L = Cm C = Moles/Liter A = KCL A = No unitC = Gram/Liter L = Cm
STEPS IN DEVELOPING A SPECTROPHOTOMETRIC ANALYTICAL METHOD • Run the sample for spectrum • 2. Obtain a monochromatic wavelength for the maximum absorption wavelength. • 3. Calculate the concentration of your sample using Beer Lambert Equation: A = KCL
There is some A vs. C where graph is linear. NEVER extrapolate beyond point known where becomes non-linear.
SPECTROMETRIC ANALYSIS USING STANDARD CURVE Avoid very high or low absorbencies when drawing a standard curve. The best results are obtained with 0.1 < A < 1. Plot the Absorbance vs. Concentration to get a straight line
CELLS UV Spectrophotometer Quartz (crystalline silica) Visible Spectrophotometer Glass IR Spectrophotometer NaCl
LIGHT SOURCES UV Spectrophotometer 1. Hydrogen Gas Lamp 2. Mercury Lamp Visible Spectrophotometer 1. Tungsten Lamp IR Spectrophotometer 1. Carborundum (SIC)
CHEMICAL STRUCTURE & UV ABSORPTION Chromophoric Group ---- The groupings of the molecules which contain the electronic system which is giving rise to absorption in the ultra-violet region.
CHROMOPHORIC STRUCTURE Group Structure nm Carbonyl > C = O 280 Azo -N = N- 262 Nitro -N=O 270 Thioketone -C =S 330 Nitrite -NO2 230 Conjugated Diene -C=C-C=C- 233 Conjugated Triene -C=C-C=C-C=C- 268 Conjugated Tetraene -C=C-C=C-C=C-C=C- 315 Benzene 261
UV SPECTROMETER APPLICATION Protein Amino Acids (aromatic) Pantothenic Acid Glucose Determination Enzyme Activity (Hexokinase)
FLUOROMETER APPLICATION Thiamin (365 nm, 435 nm) Riboflavin Vitamin A Vitamin C
VISIBLE SPECTROPHOTOMETER APPLICATION Niacin Pyridoxine Vitamin B12 Metal Determination (Fe) Fat-quality Determination (TBA) Enzyme Activity (glucose oxidase)
EXAMPLES 1. A solution of purified DNA isolated from Escherichia coli gives an absorbance of 0.793 at 260 Mm in a 1 Cm cell at pH 4.5. If E1%1Cm is 197, calculate the concentration of the solution in milligrams per milliliter. 2. Calculate the Molar Extinction Coefficient E at 351 nm for aquocobalamin in 0.1 M phosphate buffer. pH = 7.0 from the following data which were obtained in 1 Cm cell. SolutionC x 105 MIoI A 2.23 93.1 27.4 B 1.90 94.2 32.8
3. The molar extinction coefficient (E) of compound x is: 3 x 103 Liter/Cm x Mole If the absorbance reading (A) at 350 nm is 0.9 using a cell of 1 Cm, what is the concentration of compound x in sample? 4. The concentration of compound Y was 2 x 10-4 moles/liter and the absorption of the solution at 300 nm using 1 Cm quartz cell was 0.4. What is the molar extinction coefficient of compound Y? 5. Calculate the molar extinction coefficient E at 351 nm for aquocobalamin in 0.1 M phosphate buffer. pH =7.0 from the following data which were obtained in 1 Cm cell. SolutionC x 105 MI0I A 2.0 100 30
Question 6.A = 0.01E = 10000L / mole x cmL = 1cmA = ECL0.01= 10000L/mole X Cm X C (Concentration) x 1CmC = mole / LiterC = X mole / Liter = X mole (236 g/mole) / Liter (1000 Cm3) x PPM (10-6 g/Cm3)= X mole (236 g / mole) / Liter x 1 Liter / 1000 Cm3 x ( PPM) 10-6g / Cm3)=x PPMPPM = 1ug / Cm31ug = 10-6 g