Chapter 8 Electron Configurations and Periodicity
Chapter 8 Electron Configurations and Periodicity. Contents and Concepts. Electronic Structure of Atoms

Chapter 8 Electron Configurations and Periodicity
E N D
Presentation Transcript
Contents and Concepts Electronic Structure of Atoms In the previous chapter, you learned that we characterize an atomic orbital by four quantum numbers: n, l, ml, and ms. In the first section, we look further at electron spin; then we discuss how electrons are distributed among the possible orbitals of an atom. • Electron Spin and the Pauli Exclusion Principle • Building-Up Principle and the Periodic Table • Writing Electron Configurations Using the Periodic Table • Orbital Diagrams of Atoms; Hund’s Rule Copyright © Houghton Mifflin Company. All rights reserved.
Periodicity of the Elements You learned how the periodic table can be explained by the periodicity of the ground-state configurations of the elements. Now we will look at various aspects of the periodicity of the elements. • Mendeleev’s Predictions from the Periodic Table • Some Periodic Properties • Periodicity in the Main-Group Elements Copyright © Houghton Mifflin Company. All rights reserved.
Schrödinger Wave Equation! Copyright © Houghton Mifflin Company. All rights reserved.
In 1921, Otto Stern and Walther Gerlach first observed electron spin magnetism. In the diagram below, a beam of hydrogen atoms divides in two while passing through a magnetic field. This correlates with the two values of ms: +½ and -½. Copyright © Houghton Mifflin Company. All rights reserved.
The two possible spin orientations of an electron and the conventions for msare illustrated here. Copyright © Houghton Mifflin Company. All rights reserved.
An electron configuration of an atom is a particular distribution of electrons among available subshells. • An orbital diagram of an atom shows how the orbitals of a subshell are occupied by electrons. Orbitals are represented with a circle; electrons are represented with arrows up for ms= +½ or down for ms= -½. Copyright © Houghton Mifflin Company. All rights reserved.
The Pauli exclusion principle summarizes experimental observations that no two electrons in one atom can have the same four quantum numbers. • That means that within one orbital, electrons must have opposite spin. It also means that one orbital can hold a maximum of two electrons (with opposite spin). Copyright © Houghton Mifflin Company. All rights reserved.
An s subshell, with one orbital, can hold a maximum of 2 electrons. • A p subshell, with three orbitals, can hold a maximum of 6 electrons. • A d subshell, with five orbitals, can hold a maximum of 10 electrons. • An f subshell, with seven orbitals, can hold a maximum of 14 electrons. Copyright © Houghton Mifflin Company. All rights reserved.
The lowest-energy configuration of an atom is called its ground state. • Any other configuration represents an excited state. Copyright © Houghton Mifflin Company. All rights reserved.
The building-up principle (or aufbau principle) is a scheme used to reproduce the ground-state electron configurations by successively filling subshells with electrons in a specific order (the building-up order). • This order generally corresponds to filling the orbitals from lowest to highest energy. Note that these energies are the total energy of the atom rather than the energy of the subshells alone. Copyright © Houghton Mifflin Company. All rights reserved.
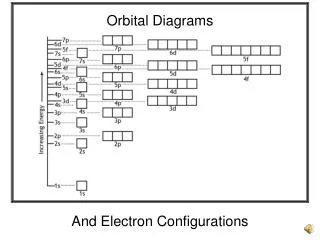
1s 2s 2p 3s 3p 3d 4s 4p 4d 4f 5s 5p 5d 5f 6s 6p 6d 7s 7p Copyright © Houghton Mifflin Company. All rights reserved.
This results in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p Copyright © Houghton Mifflin Company. All rights reserved.
Another way to learn the building-up order is to correlate each subshell with a position on the periodic table. • The principal quantum number, n, correlates with the period number. • Groups IA and IIA correspond to the s subshell; Groups IIIA through VIIIA correspond to the p subshell; the “B” groups correspond to the d subshell; and the bottom two rows correspond to the f subshell. This is shown on the next slide. Copyright © Houghton Mifflin Company. All rights reserved.
There are a few exceptions to the building-up order prediction for the ground state. • Chromium (Z=24) and copper (Z=29) have been found by experiment to have the following ground-state electron configurations: • Cr: 1s2 2s2 2p6 3s2 3p6 3d5 4s1 • Cu: 1s2 2s2 2p6 3s2 3p6 3d10 4s1 • In each case, the difference is in the 3d and 4s subshells. Copyright © Houghton Mifflin Company. All rights reserved.
There are several terms describing electron configurations that are important. • The complete electron configuration shows every subshell explicitly. • Br: 1s2 • 2s2 2p6 • 3s2 3p6 • 4s2 • 4p5 • 3d10 Copyright © Houghton Mifflin Company. All rights reserved.
The noble-gas configuration substitutes the preceding noble gas for the core configuration and explicitly shows subshells beyond that. • Br: [Ar]3d104s24p5 Copyright © Houghton Mifflin Company. All rights reserved.
The pseudo-noble-gas core includes the noble-gas subshells and the filled inner, (n – 1), d subshell. • For bromine, the pseudo-noble-gas core is • [Ar]3d10 Copyright © Houghton Mifflin Company. All rights reserved.
The valence configuration consists of the electrons outside the noble-gas or pseudo-noble-gas core. • Br: 4s24p5 Copyright © Houghton Mifflin Company. All rights reserved.
For main-group (representative) elements, an s or a p subshell is being filled. • For d-block transition elements, a d subshell is being filled. • For f-block transition elements, an f subshell is being filled. Copyright © Houghton Mifflin Company. All rights reserved.
For main-group elements, the valence configuration is in the form • nsAnpB • The sum of A and B is equal to the group number. • So, for an element in Group VA of the third period, the valence configuration is • 3s23p3 Copyright © Houghton Mifflin Company. All rights reserved.
Write the complete electron configuration of the arsenic atom, As, using the building-up principle. For arsenic, As, Z = 33. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3 Copyright © Houghton Mifflin Company. All rights reserved.
What are the electron configurations for the valence electrons of arsenic and cadmium? Arsenic is in period 4, Group VA. Its valence configuration is 4s24p3. Cadmium, Z = 30, is a transition metal in the first transition series. Its noble-gas core is Ar, Z = 18. Its valence configuration is 4s23d10. Copyright © Houghton Mifflin Company. All rights reserved.
When n = 2, there are two subshells. The s subshell has one orbital, which could hold one electron. The p subshell has three orbitals, which could hold three electrons. This would give a total of four elements for the second period. Copyright © Houghton Mifflin Company. All rights reserved.
In 1927, Friedrich Hund discovered, by experiment, a rule for determining the lowest-energy configuration of electrons in orbitals of a subshell. • Hund’s rule states that the lowest-energy arrangement of electrons in a subshell is obtained by putting electrons into separate orbitals of the subshell with the same spin before pairing electrons. Copyright © Houghton Mifflin Company. All rights reserved.
1s 2s 2p • For nitrogen, the orbital diagram would be Copyright © Houghton Mifflin Company. All rights reserved.
1s 3s 2s 4s 3d 3p 2p • Write an orbital diagram for the ground state of the nickel atom. For nickel, Z = 28. Copyright © Houghton Mifflin Company. All rights reserved.
1s 2s • Which of the following electron configurations or orbital diagrams are allowed and which are not allowed by the Pauli exclusion principle? If they are not allowed, explain why? • Allowed; excited. • p8 is not allowed. • Allowed. • d11 is not allowed. • Not allowed; electrons in one orbital must have opposite spins. • 1s22s12p3 • 1s22s12p8 • 1s22s22p63s23p63d8 • 1s22s22p63s23p63d11 Copyright © Houghton Mifflin Company. All rights reserved.
Magnetic Properties of Atoms • Although an electron behaves like a tiny magnet, two electrons that are opposite in spin cancel each other. Only atoms with unpaired electrons exhibit magnetic susceptibility. • This allows us to classify atoms based on their behavior in a magnetic field. Copyright © Houghton Mifflin Company. All rights reserved.
A paramagnetic substance is one that is weakly attracted by a magnetic field, usually as the result of unpaired electrons. • A diamagnetic substance is not attracted by a magnetic field generally because it has only paired electrons. Copyright © Houghton Mifflin Company. All rights reserved.
You learned how the organization of the periodic table can be explained by the periodicity of the ground-state configurations of the elements. Now we will look at various aspects of the periodicity of the elements. Copyright © Houghton Mifflin Company. All rights reserved.
Mendeleev’s periodic table generally organized elements by increasing atomic mass and with similar properties in columns. In some places, there were missing elements whose properties he predicted. • When gallium, scandium, and germanium were isolated and characterized, their properties were almost identical to those predicted by Mendeleev for eka-aluminum, eka-boron, and eka-silicon, respectively. Copyright © Houghton Mifflin Company. All rights reserved.
Periodic law states that when the elements are arranged by atomic number, their physical and chemical properties vary periodically. • We will look in more detail at three periodic properties: atomic radius, ionization energy, and electron affinity. Copyright © Houghton Mifflin Company. All rights reserved.
Atomic Radius • While an atom does not have a definite size, we can define it in terms of covalent radii (the radius in covalent compounds). Copyright © Houghton Mifflin Company. All rights reserved.
Trends • Within each group (vertical column), the atomic radius increases with the period number. • This trend is explained by the fact that each successive shell is larger than the previous shell. Copyright © Houghton Mifflin Company. All rights reserved.
Within each period (horizontal row), the atomic radius tends to decrease with increasing atomic number (nuclear charge). Copyright © Houghton Mifflin Company. All rights reserved.
Effective Nuclear Charge • Effective nuclear charge is the positive charge that an electron experiences from the nucleus. It is equal to the nuclear charge, but is reduced by shielding or screening from any intervening electron distribution (inner shell electrons). Copyright © Houghton Mifflin Company. All rights reserved.
Effective nuclear charge increases across a period. Because the shell number (n) is the same across a period, each successive atom experiences a stronger nuclear charge. As a result, the atomic size decreases across a period. Copyright © Houghton Mifflin Company. All rights reserved.
Atomic radius is plotted against atomic number in the graph below. Note the regular (periodic) variation. Copyright © Houghton Mifflin Company. All rights reserved.
A representation of atomic radii is shown below. Copyright © Houghton Mifflin Company. All rights reserved.
34 Se 35 Br 52 Te • Refer to a periodic table and arrange the following elements in order of increasing atomic radius: Br, Se, Te. Te is larger than Se. Se is larger than Br. Br < Se < Te Copyright © Houghton Mifflin Company. All rights reserved.
First Ionization Energy (first ionization potential) • The minimum energy needed to remove the highest-energy (outermost) electron from a neutral atom in the gaseous state, thereby forming a positive ion Copyright © Houghton Mifflin Company. All rights reserved.
Trends • Going down a group, first ionization energy decreases. • This trend is explained by understanding that the smaller an atom, the harder it is to remove an electron, so the larger the ionization energy. Copyright © Houghton Mifflin Company. All rights reserved.
Generally, ionization energy increases with atomic number. • Ionization energy is proportional to the effective nuclear charge divided by the average distance between the electron and the nucleus. Because the distance between the electron and the nucleus is inversely proportional to the effective nuclear charge, ionization energy is inversely proportional to the square of the effective nuclear charge. Copyright © Houghton Mifflin Company. All rights reserved.
Small deviations occur between Groups IIA and IIIA and between Groups VA and VIA. • Examining the valence configurations for these groups helps us to understand these deviations: • IIA ns2 • IIIA ns2np1 • VA ns2np3 • VIA ns2np4 Ittakes less energy to remove the np1 electron than the ns2 electron. It takes less energy to remove the np4 electron than the np3 electron. Copyright © Houghton Mifflin Company. All rights reserved.
These trends and reversals are visible in the graph of ionization energy versus atomic number. Copyright © Houghton Mifflin Company. All rights reserved.
The size of each sphere indicates the size of the ionization energy in the figure below. Copyright © Houghton Mifflin Company. All rights reserved.
Electrons can be successively removed from an atom. Each successive ionization energy increases, because the electron is removed from a positive ion of increasing charge. • A dramatic increase occurs when the first electron from the noble-gas core is removed. Copyright © Houghton Mifflin Company. All rights reserved.
Left of the line, valence shell electrons are being removed. Right of the line, noble-gas core electrons are being removed. Copyright © Houghton Mifflin Company. All rights reserved.