Work -- Part 1
Work -- Part 1. Physics 313 Professor Lee Carkner Lecture 6. Work. Work is force times displacement In thermodynamics we will only consider external work Involves interaction with another system or its surroundings (external to the system). Sign Conventions.

Work -- Part 1
E N D
Presentation Transcript
Work -- Part 1 Physics 313 Professor Lee Carkner Lecture 6 1
Work • Work is force times displacement • In thermodynamics we will only consider external work • Involves interaction with another system or its surroundings (external to the system) 2
Sign Conventions • Work by the system is negative • decrease internal energy • Work done on the system is positive • increase internal energy • If one system does work on the other, the sign depends on point of view 3
Work and Hydrostatic Systems • Work is not a property of the system • Neither is heat • P, V and T are • Work is a transfer of energy due to a volume change • Heat is a transfer of energy due to temperature change 4
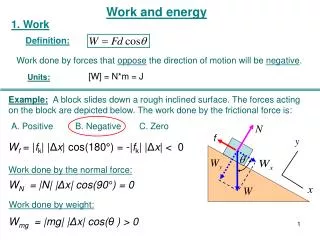
Work, Pressure and Volume W = F x dW = F dx dW = PA dx dW = -P dV • A small volume change (dV) produces a small amount of work (dW) • If dV is positive (increase in V) then W is negative (work by the system) 5
Total Work • To find the total work, integrate dW between the initial and final states: W = - P dV • Need to know P as a function of V • Equation of state • Need to limit T • W depends on both the change of volume and how the volume changed 6
PV Diagram • For a change of volume, (Vi, Pi) and (Vf,Pf) can be plotted on a PV diagram • The process by which the volume changes is a line or curve connecting the two points • The work is the area under the curve • For different processes, different curves, different amounts of work 7
Closed Cycle • If the system moves from i to f and then back to i, it is a cycle • If the same path is traveled in both directions, W=0 • if two different paths are traveled W is the area between the curves 8
Path Dependence • What are the paths? • Isothermal: keep constant T (add or subtract heat) • follow isotherm • Isobaric: constant P (add or subtract heat) • horizontal • Isochoric: keep constant volume (rigid container, W=0) • vertical 9
P-V Diagram Isobaric (p=const.) p Isothermal (T=const) Adiabatic (Q=0) Isochoric (V=const) V 10
Sign of Work • On PV diagram: • Move to right = compression = positive work • Move to left = expansion = negative work 11
Calculating Work • Need equation of state and limit on T • Example: isothermal • P is replaced with equation of state, T comes out of integral • Final expression for work in terms of constants and Vi and Vf 12
Ideal Gas: Isothermal Process PV = nRT P = nRT/V W = - (nRT/V) dV W = -nRT (1/V) dV W = -nRT ln (Vf/Vi) 13
Procedures • From equations: • Find P(V,T) • Limit T • Integrate dW • Check sign • From PV diagram: • Find area • Find sign (V increase (-) or decrease(+)) 14