Electrolytes
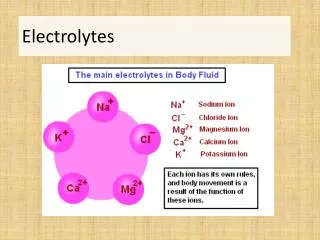
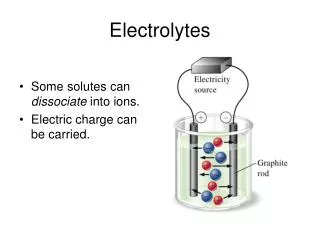
Electrolytes. Part 1. Cathode. Anode. Electrolytes. Electrolytes are ions capable of carrying an electricl charge Anions: (-) → Anode Cations: (+) → Cathode Major cations of the body Na + , K + , Ca +2 & Mg +2 Major anions of the body Cl - , HCO 3 - , HPO 4 -2 & SO 4 -2.

Electrolytes
E N D
Presentation Transcript
Electrolytes Part 1
Cathode Anode Electrolytes • Electrolytes are ions capable of carrying an electricl charge • Anions: (-) → Anode • Cations: (+) → Cathode • Major cations of the body • Na+, K+, Ca+2 & Mg+2 • Major anions of the body • Cl-, HCO3-, HPO4-2 & SO4-2 M. Zaharna Clin. Chem. 2009
Essential Component in Numerous processes • Volume and osmotic pressure (Na+, K+, Cl-) • Myocardial rhythm and contraction (K+, Mg2+, Ca2+) • Cofactors in enzyme activation (Mg2+, Ca2+, Zn2+). • Regulation of ATPase ion pump (Mg2+) • Acid/Base balance (pH) (HCO3-, K+, Cl-) • Coagulation (Mg2+, Ca2+) • Neuromuscular (K+, Mg2+, Ca2+) The body has complex systems for monitoring and maintaining electrolyte concentrations M. Zaharna Clin. Chem. 2009
Maintenance of water homeostasis is vital to life for all organisms • Maintenance of water distribution in various body fluids is a function of electrolytes (Na+, K+, Cl- & HCO3-) M. Zaharna Clin. Chem. 2009
Water • Average water content of human body is 40-75% of total body weight. • Solvent for all body processes • Transport nutrients to cells • Regulates cell volume • Removes waste products → urine • Body Coolant → sweating • Water is located in intracellular and extracellular compartments M. Zaharna Clin. Chem. 2009
Water • Normal plasma ~ 93 % H2O, the rest is mixture of Lipids and proteins. • Concentration of ions within the cells and plasma is maintained by: • Energy consumption: Active transport • Diffusion: Passive transport • Maintaining conc. of electrolytes affect distribution of water in compartments • Most membranes freely permeable to water • Conc. of ions on one side affect flow of water across the membrane M. Zaharna Clin. Chem. 2009
Osmolality • Physical property of a solution based on the concentration of solutes per kilograms of solvent. (mOsm/Kg) • Sensation of thirst & arginine vasopressin hormone (AVP) [formerly, Antidiuretic hormone (ADH)] are stimulated by hypothalamus in response to increased blood osmolality • Thirst → more water intake • AVP → increase water absorption in kidney M. Zaharna Clin. Chem. 2009
Clinical Significance • Osmolality is the parameter to which hypothalmus responds to maintain fluid intake. • The regulation of osmolality also affects the Na+ concentration in plasma • 90% of osmotic activity in plasma • Another process affects Na+ concentration is regulation of blood volume. M. Zaharna Clin. Chem. 2009
Clinical significance • To maintain normal plasma osmolality (275-295 mOsm/Kg) hypothalamus must respond quickly to small changes • 1-2% increase in osmolality: 4 fold increase in AVP secretion. • 1-2% decrease in osmolality: shuts off AVP secretion. • Renal water regulation by AVP and thirst play important roles in regulating plasma osmolality. • Renal water excretion is more important in controlling water excess, • whereas thirst is more important in preventing water deficit or dehydration. • Consider what happens in several conditions. M. Zaharna Clin. Chem. 2009
Water Load • Excess intake of water lower plasma osmolality • Kidney is important in controlling water excess • AVP and thirst are suppressed • Water is not reabsorbed, causing a large volume of dilute urine to be excreted • Hypoosmolality and hyponatremia usually occur in patients with impaired renal excretion of water M. Zaharna Clin. Chem. 2009
Water deficit • As a deficit of water, plasma osmolality begins to increase • Both AVP secretion and thirst are activated. • Although AVP contributes by minimizing renal water loss, thirst is the major defense against hyperosmolality and hypernatremia. • A concern in infants, unconscious patients, or anyone who is unable to either drink or ask for water M. Zaharna Clin. Chem. 2009
Regulation of blood volume • Blood volume essential in maintaining blood pressure and ensure perfusion to all tissue and organs. • Regulation of both sodium & water are interrelated in controlling blood volume • Renin-angiotensin-aldosterone: system of hormones that respond to decrease in blood volume and help maintain the correct blood volume. M. Zaharna Clin. Chem. 2009
Regulation of blood volume • Changes in blood volume detected by receptors in: • the cardiopulmonary circulation , • carotid sinus, • aortic arch • and glomerular arterioles • They activate effectors that restore volume by: • appropriately varying vascular resistance, • cardiac output, • and renal Na and H2O retention. M. Zaharna Clin. Chem. 2009
Angiotensin converting enzyme (ACE) M. Zaharna Clin. Chem. 2009
Regulation of blood volume • Other Factors effecting blood volume: • Atrial natriuretic Peptide (ANP) → sodium excretion → ↓ blood volume • Volume receptors → release of AVP → conserve water → ↑ blood volume • Glomerular filtration rate (GFR) ↑ in volume expansion and ↓ in volume depletion M. Zaharna Clin. Chem. 2009
Determination of Osmolality • Serum or urine sample (plasma not recommended due to the use of anticoagulants) • Based on properties of a solution related to the number of molecules of solutes per kilogram of solvent such as: • Freezing point • Vapor pressure M. Zaharna Clin. Chem. 2009
Determination of Osmolality • Freezing Point Osmometer: • Standardized method using NaCl reference solution. • Specimen is supercooled to -7ºC, to determine freezing point. • ↑ osmolality causes depression in the freezing point temp. • More solutes present the longer the specimen will take to freeze. M. Zaharna Clin. Chem. 2009
OsmolalGap • Osmolal gap is the difference between the measured osmolality and the calculated one. Osmolal Gap= measured osmolality - calculated osmolality • The osmolal gap indirectly indicates the presence of osmotically active substances other than sodium, urea or glucose. (ethanol, methanol or β-hydroxybutyrate) M. Zaharna Clin. Chem. 2009
Sodium • Most abundant extracellular cation- 90% • Major function is maintaining the normal water distribution & osmotic pressure of plasma • Role in maintaining acid-base balance (Na+, H+ exchange mechanism) • Normal range Serum: 136-145 mmol/L • ATPase ion pump: the way the body moves sodium and potassium in and out of cells. • 3 Na+ out of the cell for every 2 K + in and convert ATP to ADP. M. Zaharna Clin. Chem. 2009
Regulation of Sodium Balance • Plasma Na+ concentration depends: • on the intake and excretion of water • and, on the renal regulation of Na+ • Three processes are of primary importance: • intake of water in response to thirst (p. osmolality) • the excretion of water (AVP release) • the blood volume status, which affects Na+ excretion through aldosterone, angiotensin II, and ANP (atrialnatriuretic peptide). M. Zaharna Clin. Chem. 2009
Regulation of Sodium Balance • 70 % of sodium that is filtered is reabsorbed in proximal tubules. • Remainder occurs in the ascending loop of Henle (without water absorption) & DCT under regulation of Aldosterone • Renin-Angiotensin system • Atrial natriuretic Peptide (ANP) → sodium excretion M. Zaharna Clin. Chem. 2009
Hyponatremia • Defined as a serum/plasma level less than 135 mmol/L. • One of the most common electrolyte disorders in hospitalized and non-hospitalized patients • Levels below 130 mmol/L are clinically significant. M. Zaharna Clin. Chem. 2009
Hyponatremia M. Zaharna Clin. Chem. 2009
Hypernatremia • Hypernatremia: increased sodium concentration > 145 mmol/l • Result of excess water loss in the presence of sodium excess, or from sodium gain M. Zaharna Clin. Chem. 2009
Sodium determination • Methods: • Flame emission spectrophotometry • Atomic absorption spectrophotometry • Ion Selective electrode M. Zaharna Clin. Chem. 2009
Atomic absorption spectrophotometry M. Zaharna Clin. Chem. 2009
Ion Selective electrode M. Zaharna Clin. Chem. 2009
Potassium • Major intracellular cation • 20 X greater concentration in the cell vs. outside. • 2% of the bodies potassium circulates within the plasma. • Function: • Regulates neuromuscular excitability • Hydrogen ion concentration • Intracellular fluid volume M. Zaharna Clin. Chem. 2009
Effects on Cardiac muscle • Ratio of K+ intracellular & extracellular is important determinant of resting membrane potential across cell membrane • Increase plasma potassium; decreasing the resting membrane potential, increase excitability, muscle weakness • Decrease extracellular potassium; decrease excitability M. Zaharna Clin. Chem. 2009
Potassium Role in Hydrogen Concentration • In hypokalemia (low serum K+), • As K+ is lost from the body, Na+ and H+ move into the cell. • The H+ concentration is, therefore, decreased in the ECF, resulting in alkalosis. M. Zaharna Clin. Chem. 2009
Regulation of potassium • The kidneys are important in the regulation of K+ balance. • Initially, the proximal tubules reabsorb nearly all the K+. • Then, under the influence of aldosterone, K+ is secreted into the urine in exchange for Na+ in both the distal tubules and the collecting ducts. • Thus, the distal tubule is the principal determinant of urinary K+ excretion. • Most individuals consume far more K+ than needed; the excess is excreted in the urine but may accumulate to toxic levels if renal failure occurs. M. Zaharna Clin. Chem. 2009
Hypokalemia • Decrease of serum potassium below 3.5 mmol/l M. Zaharna Clin. Chem. 2009
Hyperkalemia • Increase potassium serum levels > 5 mmol/l • Associated with diseases such as renal and metabolic acidosis M. Zaharna Clin. Chem. 2009
Potassium determination Assay method: • Ion selective Electrode • a valinomycin membrane is used to selectively bind K+ M. Zaharna Clin. Chem. 2009
Chloride • Major extracellular anion • Cl– is involved in maintaining: • osmolality, • blood volume, • and electric neutrality. • In most processes, Cl– ions shift secondarily to a movement of Na+ or HCO3–. • Cl– ingested in the diet is Completely absorbed by the intestinal tract. M. Zaharna Clin. Chem. 2009
Chloride • Cl– ions are filtered out by the glomerulus and passively reabsorbed, in Conjuction with Na, by the proximal tubules. • Excess Cl– is excreted in the urine and sweat. • Excessive sweating stimulates aldosterone secretion, which acts on the sweat glands to Conserve Na and Cl–. M. Zaharna Clin. Chem. 2009
Electric Neutrality • Sodium/chloride shift maintains equilibrium within the body. • Na reabsorbed with Cl in proximal tubules. • Chloride shift • In this process, carbon dioxide (CO2) generated by cellular metabolism within the tissue diffuses out into both the plasma and the red cell. • In the red cell, CO2 forms carbonic acid (H2CO3), which splits into H+ and HCO3- (bicarbonate). • Deoxyhemoglobin buffers H+, whereas the HCO3- diffuses out into the plasma and Cl- diffuses into the red cell to maintain the electric balance of the cell M. Zaharna Clin. Chem. 2009
Hypochloremia • Hypochloremia: < 98 mmol/l M. Zaharna Clin. Chem. 2009
Hypercholremia • Hypercholremia: > 109 mmol/l M. Zaharna Clin. Chem. 2009
Assay • Coulometric titration (ref. method) • Ion selective electrode M. Zaharna Clin. Chem. 2009