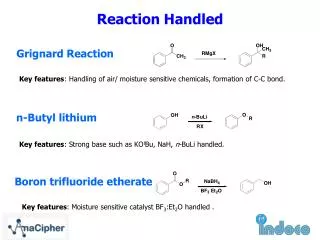
GRIGNARD REAGENTS
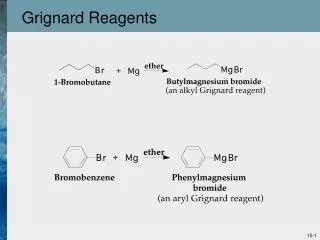
GRIGNARD REAGENTS. ORGANOMETALLICS FROM DIVALENT METALS. Formation of Grignard Reagents. This reaction was discovered by the Frenchman, Victor Grignard -- Nobel Prize in 1912. HALIDE REACTIVITIES. RX + Mg R-Mg-X. RELATIVE RATES :.

GRIGNARD REAGENTS
E N D
Presentation Transcript
GRIGNARD REAGENTS ORGANOMETALLICS FROM DIVALENT METALS
Formation of Grignard Reagents This reaction was discovered by the Frenchman, Victor Grignard -- Nobel Prize in 1912
HALIDE REACTIVITIES RX + Mg R-Mg-X RELATIVE RATES : R-I > R-Br > R-Cl FAST SLOW expensive, good compromise less reactive, but easy to prepare or buy not readily available
FORMATION OF A GRIGNARD REAGENT + ether Ethers are obligatory solvents for the Grignard Reaction. The reaction doesn’t work without an ether solvent. Typical ether solvents are: Diethyl ether (b.p. 35o C) Tetrahydrofuran (b.p. 65o C) Dioxane (b.p. 101o C) THF
Why might you need different solvents? Consider two different leaving groups: Bromine is more reactive than chlorine. The second reaction is too slow at 35 °C, therefore we use a higher boiling solvent.
Ethers stabilize the Grignard Complex Formation of this complex is exothermic; the reaction is sufficiently exothermic to boil the solution without having to add external heat!
3s 3p 3s 3p 3s 3p ETHERS COORDINATE INTO EMPTY 3p ORBITALS Mg: [Ne]3s2 promotion . . Mg . . +RX R X two bonds form R-Mg-X two ethers coordinate
Et Et .. O .. Et .. : O Et Ethers stabilize the Grignard Complex ether molecules coordinate into empty 3p orbitals on magnesium THE GRIGNARD COMPLEX R Mg X
R-Mg-X IS A USEFUL SIMPLICATION CHEMISTS HAVE BEEN ARGUING ABOUT THE STRUCTURE OF THE GRIGNARD REAGENT FOR YEARS AND STILL CANNOT AGREE The complete structure of the Grignard reagent is quite complex. It is probably an equilibrium mixture of the type: ….. and the actual reactive species may be a complex dimer. While this complex picture may be more correct, it is easier to treat the Grignard reagent as if it were simply R-Mg-X, which is what we shall do in this course.
We can view the Grignard as a hybrid reagent. RMgX is a source of a carbanion ( R:- ) just like RLi. Therefore we expect Grignard reagents to be both a strong base and good nucleophile.
d- d+ - : PERCENT IONIC CHARACTER carbanion + covalent ionic most reactive bond percent ionic strongest base C-K 51 C-Na 47 C-Li 43 C-Mg 35 C-Zn 18 C-Cd 15 C-Cu 9 Less ionic character than organolithium compounds. More covalent. best nucleophile least reactive
Grignard Reagents give all the same reactions as alkyllthium compounds. R-Mg-X R-Li
Grignard reagents are strong bases and react readily with any slightly acidic hydrogen ( compare R-Li). Any source of H+ will bring about this reaction: water acids alcohols amines alkynes carboxylic acids atmosperic moisture Any -O-H, -S-H, or -N-H bonds are sufficiently acidic to react.
A DELIBERATE SOURCE OF PROTONS OR DEUTERIUM R ( or H2O) This is not a stereospecific synthetic method. R+S
A A C C : : B B C C INVERSIONS OF THE GRIGNARD REAGENT Grignard reagents lose stereochemistry (racemize) when made from an enantiomerically pure alkyl halide. A * C X B C chiral halide MgX+ scrambles stereochemistry Organolithium compounds also usually loose stereochemistry, however, some RLi compounds retain configuration at low temp- eratures (-60 oC or below), where the rate of inversion is slow.
SYNTHESIS OF ALCOHOLS AND CARBOXYLIC ACIDS
Reaction with Carbonyl Compounds Just as with alkyllithiums ( RLi ): • formaldehyde primary alcohols • other aldehydes secondary alcohols • ketones tertiary alcohols • carbon dioxide carboxylic acids Read the following section only from Chap 16 (16.7)
REACTION WITH CARBON DIOXIDE O ether R-Mg-X + O=C=O R-C-O- MgX+ H3O+ R-COOH (s) 1) CO2 Mg ether 2) H3O+
STARTING HALIDES YOU CANNOT USE
NOTE TO STUDENTS CAUTION Most Grignard and Alkyllithium compounds are not stable compounds. You cannot buy them, and you must make them and use them immediately. Therefore, I will expect you to show the reaction that forms these reagents in any synthesis problem. Do not “pull a Grignard or Alkyllithium out of your hat” you must show how it is made from a halide. Mg etc RX RMgX (CH3)2C=O ether Also ….. do not forget the hydrolysis step with H3O+
THESE REACTIONS DON’T WORK …. WHY NOT? Mg ether Mg ether Mg ether INTERFERING GROUPS !