Plant Biotechnology
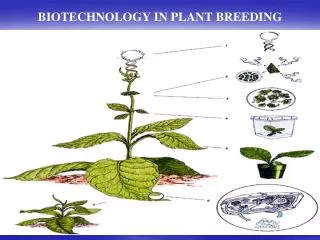
Plant Biotechnology. Two broad categories New techniques for plant propagation Already a profitable enterprise Works with existing germplasm Mutation breeding The creation of transgenic plants. The Creation Of Transgenic Plants. The introduction and expression of novel genes in plants

Plant Biotechnology
E N D
Presentation Transcript
Plant Biotechnology • Two broad categories • New techniques for plant propagation • Already a profitable enterprise • Works with existing germplasm • Mutation breeding • The creation of transgenic plants
The Creation Of Transgenic Plants • The introduction and expression of novel genes in plants • Creates new germplasm
Transgenic Plant Biotechnology • Made possible about 30 years ago by the development of ways to introduce and express foreign genes in plants. • Commercial ventures have been funded by • Chemical companies; Dupont (Pioneer), Monsanto, Dow, Syngenta (Zeneca, Novartis), BASF, Bayer(Aventis) • Seed Companies; Pioneer, Delta-Pine Land, Seminis • Venture Capital has funded many smaller companies.
Endless Possibilities • The ability to express any gene in any part of a plant means that the possibilities for crop improvement are theoretically limitless. • In reality many potential improvements are beyond our present capabilities. • Pleitropic effects are often a fatal flaw
Disease Resistance: Practicalities • It’s easy to create a disease resistant transgenic plant. • It’s very hard to make one that is commercially viable
Why can’t famers buy this product? • Don’t know for sure • Didn’t work in larger scale field trials? • Didn’t work well enough to make it economically viable? • Other issues (toxicity etc.)?
Issues • Need a suitable gene and promotor • Legal issues • Ability to transform plant • Technical • Legal • Pleiotropic Effects • Yield depression • Economic Impact • Does it save the farmer money or time? • Does it increase yield or obviate the need for fungicide applications? • And/or would consumers be willing to pay more for it? • Regulatory • Allergenic, toxic • weedy • Public Perception
Requirements -1 • Techniques for the introduction of DNA into plants. Available for most plants. • Agrobacterium • Particle Bombardment
Agrobacterium • Agrobacterium tumefaciens cause tumorous growth on plants. • Does this by transferring large extra-chromosomal piece of DNA (T-DNA) into host genome. T-DNA encodes several “oncogenes”.
Agricultural biotechnology: Gene exchange by design Stanton B. Gelvin Nature 433, 583-584(10 February 2005)
Agricultural biotechnology: Gene exchange by design Stanton B. Gelvin Nature 433, 583-584(10 February 2005)
Biolistics • DNA is bound to metal (often gold) particles and literaly shot into the cell (gunpowder or compressed gas). • The rare events in which foreign DNA has been incorporated into the host genome are identified using screening for selectable marker phenotypes (e.g. herbicide resistance)
http://www.plant.uoguelph.ca/research/homepages/raizada/Equipment/RaizadaWeb%20Equipment%20Images/8.%20PDS100HE%20gun.jpghttp://www.plant.uoguelph.ca/research/homepages/raizada/Equipment/RaizadaWeb%20Equipment%20Images/8.%20PDS100HE%20gun.jpg
Requirements-2 • Components for expressing a gene in a plant Gene: Codes for protein. Can also be designed to turn off an endogenous gene Promoter: Controls when where and how much gene is turned on. For disease resistance we might want a disease or pathogen-inducible promoter Terminator: Needed at the end of a gene
Requirements-3 • Access to the appropriate intellectual property • Transformation techniques • IP specific to plant or class of plant • Genes, promoters, selectable markers etc. • Approaches -e.g. RNAi
Problems-1 • Genetic engineering can be unpredictable • Somewhat analogous to traditional plant breeding- dealing with a very complex situation • Every gene/plant combination is unique. Every transformed plant is unique- need to screen through lots of transformants to find a “good one” • Results seen in the laboratory often cannot be replicated under field conditions.
Problems-2 • Intellectual property is an extremely complex area • Often several different pieces of IP involved in a single transgenic plant • Includes IP on the transformation method, each element of the expression construct • Especially difficult for smaller companies
Problems-3 • Very strict regulations • Transgenic plants need to go through an extremely stringent process of analysis. • Regulated by USDA, EPA and FDA • The exact position of all the inserted DNA has to be documented. • Difficult to prevent pollen spread in certain cases • E.g. Prodigene case • A large problem for BioPharming
Ultimately this regulation is probably a good thing for the industry but it creates a LARGE FINANCIAL HURDLE • ~~$60m to create a commercial transgenic plant
NY TIMES Spread of Gene-Altered Pharmaceutical Corn Spurs $3 Million Fine By ANDREW POLLACK Published: December 7, 2002 A biotechnology company will pay the government about $3 million to settle charges that it did not take proper steps to prevent corn that was genetically engineered to produce pharmaceuticals from entering the food supply. Aventis Says More Genetically Altered Corn Has Been Found By DAVID BARBOZA Published: November 22, 2000 The Aventis Corporation said yesterday that a genetically altered protein unapproved for human consumption had been discovered in a variety of corn that could be headed toward the nation's food supply.
Problems-5 • Development of product is very expensive. • Transgenic plant needs to be equal of elite varieties for all other traits. • New germplasm may supercede variety into which construct has been introduced
Problems-6 • Public acceptance • Most criticisms are unsubstantiated but public is skeptical. • European restrictions mean it’s hard for US farmers to export • Labeling requirement, although ostensibly fair, is tantamount to a complete ban
Approaches to Genetic Engineering for Disease Resistance • Commercially available virus and insect resistant plants are available • No commercially available Fungal or bacterial resistant plants (see above!)
Possible Approaches • Using R-genes • Many R-genes cloned • Could simply introduce cloned R-gene to a new species or line by transformation. • This works in some cases but: • R-genes tend to be quite specific and easily overcome • Often don’t work in other species anyway • This might be a good way to rapidly pyramid R-genes or to introduce different alleles of the same gene into a single background
Using R-genes continued • R-gene expression can cause growth reductions • E.g Rpm1 • This is likely to be a case-by-case issue though and may be remedied by using weaker promoters
Using R-genes- continued • R-genes provide very effective resistance when triggered. The trick is to trigger them at the right time and place. • How about if you could activate R-genes at an appropriate time when the plant is attacked by a broad variety of pathogens? • Overcomes the disease/race-specificity problem
This approach is dependent of finding appropriate promoters. • Genomics approaches are ideal for finding such promoters
Using R-genes- Custom Design • Can you design an R-gene to interact with and be activated by your molecule of choice? • Ideally it would interact with a molecule essential for fungal pathogenicity • This would mean the fungus would incur a large penalty if it evolved to overcome the R-gene.
Directed molecular evolution- Gene Shuffling • Lassner and Bedbrook Current Opinion in Plant BiologyVolume 4, Issue 2, 1 April 2001, Pages 152-156
Gene shuffling has been used to identify recognition determinants in R-genes • Wulff BBH, Thomas CM, Smoker M, Grant M, Jones JDG: Domain swapping and gene shuffling identify sequences required for induction of an Avr-dependent hypersensitive response by the tomato Cf-4 and Cf-9 proteins. Plant Cell 2001, 13:255-272. • Attempts have been made to shuffle for specific novel recognition • No success (that I know of). • This would really be the dream scenario if possible.
Using R-genes- finding novel R-genes from other species • See Wroblewski et al Plant Phys 2009 150:1733-1749. • Identified and cloned 171 putative bacterial effector genes, from a variety of bacterial pathogens, using a set of predictive rules. • Expressed them in 59 different plant genotypes including accessions of lettuce, tomato, pepper, Arabidopsis, tobacco • Used an agrobacterium transient assay • 10,089 different combinations.
More than a third elicited a reaction in at least one genotype
Can express effectors in plant pathogens infecting the target host • Can’t do transient agro infection in all plants • Sohn et al 2007 Plant cell 19:4077-4090 • Expressed effectors from H. parasitica in P. syringae and got appropriate response in the host.
So what? • The thought here is: • In the target plant/pathogen system; Identify effectors that are widespread within the pathogen species. These are likely important for pathogenesis • By expressing these effector genes in heterologous hosts can find R-genes that recognize them. • Clone the R-genes and move them into target host
There R-genes stand a chance of being durable since they target conserved effectors that are likely to be hard for the pathogen to lose.
This is essentially what was done in: • Vleeshouwers, et al. 2008. Effector genomics accelerates discovery and functional profiling of potato disease resistance and Phytophthora infestans avirulence genes. PLoS ONE, 3:e2875 • Expressed 54 predicted P. infestans effector genes in a set of wild potatoes using a PVX system • Found a couple of R-genes
Gene Silencing -RNAi • A technology that uses a basic plant process to silence selected genes From Wikipedia
RNAi • This has been used extensively to silence plant genes. • This the basis of most transgenic viral resistance in plants. • Viral protein gene is silenced in plant cells • Virus can’t reproduce, spread • A transgenic success story (more-or-less)
Genetic Engineering Success Stories Transgenic Papaya, resistant to Papaya Ring Spot
RNAi • What about other uses of RNAi • RNAi is a process common to most higher organisms including plants, fungi, insects, vertebrates. • Can you get the small RNAs into Fungi, nematodes, bacteria to silence important genes? • In some cases yes
Does RNAi have broader applications for Transgenic Disease Resistance? • Fungi/Oomycetes Resistance to Phytophthora nicotianae in Tobacco www.venganzainc.com
Insects • Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol • Nature Biotechnology 25, 1307 - 1313 (2007) • Control of coleopteran insect pests through RNA interference • Nature Biotechnology 25, 1322 - 1326 (2007)
Nematodes • Engineering broad root-knot resistance in transgenic plants by RNAi silencing of a conserved and essential root-knot nematode parasitism gene • PNAS | September 26, 2006 | vol. 103 | no. 39 | 14302-14306
Trends in BiotechnologyVolume 25, Issue 3, March 2007, Pages 89-92
This is a really great approach if it works • Very specific • Can target essential processes so very difficult to overcome • No growth penalty for plant
Other Approaches to Engineering Disease Resistance in Plants • Manipulate components of the existing defence reponse • Introduced preformed anti-fungal metabolites from other species • Also could manipulate cell-death pathways
Existing Defence Response • Can try to manipulate single defensive proteins • E.g Anti Fungal Peptides (Defensins), PR proteins • Or can try to work with “Master Switches” • Genes that effect the expression or function of whole suites of other genes • Kinases, Transcription Factors and other signalling molecules
There are numerous examples of both these approaches ‘working’ in the literature • E.g NPR1 overexpression • Generation of broad-spectrum disease resistance by overexpression of an essential regulatory gene in systemic acquired resistance • Proc Natl Acad Sci U S A. 1998 May 26; 95(11): 6531–6536. • But growth peanlties are frequently observed