Ch 8 Main Group Elements
Ch 8 Main Group Elements. General Trends Introduction Twenty top industrial chemicals are main group elements or compounds 8 of the top 10 are “inorganic” 9 of the top 20 are “organic” Provide context for concepts also important later Clear up some misconceptions C with > 4 bonds

Ch 8 Main Group Elements
E N D
Presentation Transcript
Ch 8 Main Group Elements • General Trends • Introduction • Twenty top industrial chemicals are main group elements or compounds • 8 of the top 10 are “inorganic” • 9 of the top 20 are “organic” • Provide context for concepts also important later • Clear up some misconceptions • C with > 4 bonds • Alkali metal anions • Noble Gas compounds
Physical Properties • Main group elements characterized by s/p electrons following the octet rule • Metals give up electrons to achieve filled valence shell • Nonmetals accept electrons to achieve filled valence shell • Conductance • Metals have loosely bound e- so they can conduct electricity Electron Sea Model b) Nonmetals have localized lone pairs = insulators c) Metalloids/Semimetals • Diagonal from B to Po • Can often be intrinsic or doped semiconductors Resistivity
3) Electronegativity a) F most electronegative; trend is to decrease down and to left in periodic table b) H has different value than other Group I elements: duet rule c) Noble Gases calculated to be more electronegative than halogens i. Smaller size due to larger Z ii. Should pull on shared e- quite strongly
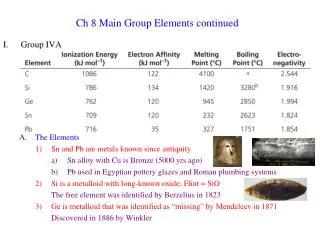
Ionization Energy • E ----> E+ + e- • Trends similar to electronegativity with a few exceptions • B < Be because Be has filled subshell 2s2 and B easily loses 2s22p1 • O < N because N has half-filled subshell 2s2p3 and O easily loses 2s22p4
Chemical Properties • Elements within a group have similar reactivities because of same valence • Diagonal (upper left to lower right) have some similarities • Electronegativity about the same: B-Si-As-Te all between 1.9-2.2 • Solubility: LiF, MgF2 sparingly soluble • Size and electronic structure probably most important • First Row Anomaly for Li through Ne • Significant differences from rest of their groups • F2 much weaker bond than extrapolated from I2, Br2, and Cl2 • HF weak acid while HI, HBr, and HCl are strong acids • C=C, C≡C prevalent, while Si=Si and Si≡Si very rare • Strong H-bonding for all first row elements • Result of small size and high Electronegativity • Hydrogen • Position in Periodic Table • 1s1 electron configuration is like Alkali Metals but not chemically similar • One e- short of octet is like Halogens, but has limited similarity H2 like X2 H- forms like X-
What about placing H in Group IVA? • Half-filled valence shell • Electronegativity is about the same • Covalent bond formation favored over ionic • Best to Treat H as unique, not part of any other Group • Abundance • Most abundant element in the universe; found mostly in stars and in “space” • 3rd most abundant element on Earth’s Surface; found mostly in compounds (H2O) • Isotopes • Hydrogen = 1H = 99.9844% • Deuterium = 2H = D = used in NMR solvents and “isotope effect” studies • Tritium = 3H = T • Radioactive with 12.35 y half-life: • Produced in Nuclear Reactors: • Used as tracer for H absorption; ground water flow detection
Chemical Properties • Gains e- to achieve Noble Gas configuration = Hydride = H:- • Ionic compounds analogous to halides: NaH, KH, etc... • Covalent C—H bonds (consider 2 e- in bond as making H into a hydride) • Lone pair donor (Lewis Base) functioning as Ligand to metals: Li2[NiH4] • Organic Reducing agent: NaBH4, LiAlH4 • Loses one e- to become H+ = Proton • Always associated with other molecules due to small size: H3O+ • Basis for one definition of Acids/Bases • Can form Hydrogen Bonds • Combustion • H2 + ½ O2 ----> H2O • Pollutant free energy source? • Problems: • Generating cheap H2 • Currently, most H2 comes from natural gas (generates CO2) • Ideally, need cheap way to generate from water • Storage is also a problem: metals? carbon nanotubes?
Group IA Alkali Metals • The Elements • Known since antiquity in salts (NaCl) used for food flavoring and preservation • K, Na essential for human life • Pure elements discovered “recently” due to difficulty in reduction of cations • 1807 Davy discovered K, Na from electrolysis of molten KOH, NaOH • 1817 Davy discovered Li from Li2O • Cs, Rb discovered 1860, 1861 using newly invented spectroscope • Fr discovered in 1939 as a radioactive isotope from the decay of Actinium • Physical Properties • Silvery metals that are highly reactive and have low mp • Stored under oil to prevent reaction with water in air • Soft enough to cut with a knife by hand
Chemical Properties • Easy loss of 1 e- (lowest IE of all the elements) • Excellent reducing agents (get oxidized themselves) • 2 Na + 2 H2O ----> 2 NaOH + H2 Exothermic • Can form oxides (M2O), peroxides (M2O2), and superoxides (MO2) • Dissolve in NH3(l) to give solvated e- • Na + x NH3 ----> Na+ + e(NH3)x- • Even better conductor than salt water, about as good as a molten metal • Paramagnetic because of unpaired e- • Less dense than NH3(l) because e- forms cavities in solution • Blue color at low conc. (e-); coppery color at high conc. (M-) • Excellent reducing agents • Slowly decompose to form MNH2 + ½ H2 • M+ forms Lewis Acid/Lewis Base Complexes • Crown Ethers = cyclic –[CH2CH2O]- polyethers donate lone pairs from O • Size selective for specific alkali metal cation • HSAB argument: hard O and hard alkali metal cation
Crown Ether, Cryptand and Complex Formation Constants of Cryptands with Alkali Metal Cations
Alkalides = M- (James Dye) • 2 Na + [2.2.2]Cryptand ----> Na([2.2.2]Cryptand)+ + Na- • Disproportionation: 2 Na ----> Na+ + Na- • Alkalides have an s2d10 electron configuration • Very powerful reducing agents • Usually unstable and decompose quickly even at room temperature • Group IIA Alkaline Earths • The Elements • Ca and Mg are abundant and have been known since antiquity • CaO = lime used by Romans in brick mortars • CaSO4• 2 H2O = gypsum used by Egyptians in plasters • Mg is strong, light construction material • Ca found in bone, teeth; other biological functions • Sr and Ba less abundant, but also found as Sulfates and Carbonates • Be even less abundant, found in Beryl = Be3Al2(SiO3)6 = emerald, aquamarine • used in alloys with Cu, Ni, etc… • Reduces corrosion • Ra is radioactive, discovered in 1898 by Marie Curie
Smaller than Group IA elements due to larger Z • More dense • Higher I.E. • Higher mp, bp, DHfus, DHvap • Chemical Properties • Tend to lose 2 e- and are thus good reducing agents • Mg + 2 H+ ----> Mg2+ + H2 • More reactive down group: Ca + 2 H2O ----> Ca(OH)2 + H2 • Be is unique: primarily covalent bonds rather than ionic; also extremely toxic
Grignard Reagents = RMgX • Complex structures • RMgX + H2C=O ----> RCH2OH • Photosynthesis depends on Mg • Chlorophyll contains Mg in a macrocycle • Complex chain of reactions to produce sugars from light, CO2 • Cement = complex mixture of Ca silicates, aluminates, and ferrates • Most important construction material (1021 kg/yr worldwide) • With H2O and sand, concrete is formed • H2O and OH- link the other components into large, strong crystals • Group IIIA • Boron • Nonmetal chemically most similar to C and Si • Hydride formation like C • Borates = Oxygen containing minerals like Silicates • Pure element has many allotropes, usually containing B12 icosahedral units
Boranes = BxHy compounds • H often bridges B atoms in boranes • Diborane = B2H6 • 12 valence electrons, 8 involved in terminal B—H bonds • 4 electrons in two 3-center, 2-electron bonds B—H—B • Group Theory analysis of D2h symmetry produces Ag and B1u bonding Molecular Orbitals
Carboranes = CxByHz compounds also have bridging alkyl groups • Bridging also seen in Al compounds • Isotopes • 11B (80.4%) and 10B (19.6%) • 10B high neutron absorption (M.F. Hawthorne) • Used in cancer therapy • B compounds localize in cancer cells • Irradiate with neutrons • Kill cancer cell specifically
Metallic Group IIIA Elements • Formation of 3-coordinate Lewis Acids • BX3, AlX3 excellent Lewis Acids: AlX3 + RX ----> R+ + AlX4- • Accept lone pair form Lewis Base to fill octet and become 4-coordinate • Usually form 3+ cations • Inert Pair Effect = metal with oxidation state 2 less than valence number • Tl ----> Tl+ + e- (Also seen in other metals: Pb ----> Pb2+ + 2 e-) • Retains s electrons because entirely filled subshell is somewhat stabilized • Explanation is actually more complex
Parallels to Organic Chemistry • Borazine = inorganic benzene = B3N3H6 • Chemistry is different even though structure is similar • Polar B—N bond • Nu attack N; E attack B more easily than in Benzene • BN = boron nitride • Diamond-like structure is hard like diamond • Graphite Structure is poor conductor because less delocalized electrons