HRP-12: A Novel HSP;Its Distribution and Function.
330 likes | 680 Views
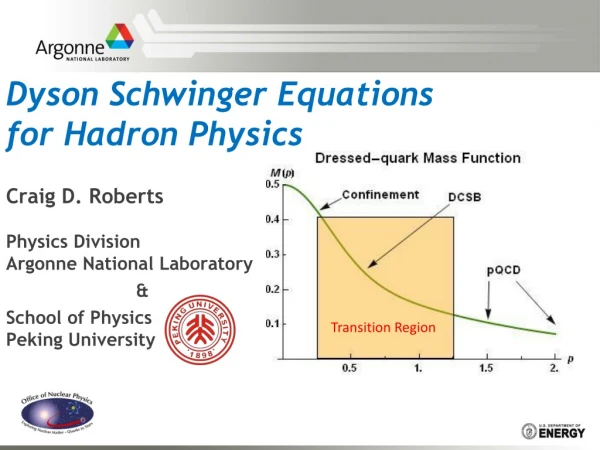
HRP-12: A Novel HSP;Its Distribution and Function. Syed Muzamil H Zaidi. PGY-3 Mentor: Dr S. Jayanth Samuel M.D., Ph.D VAMC Buffalo,NY. Heat Shock Proteins. A group of molecular Chaperones. Chaperones are molecules mostly proteins with diverse functions especially protein folding.

HRP-12: A Novel HSP;Its Distribution and Function.
E N D
Presentation Transcript
HRP-12: A Novel HSP;Its Distribution and Function. Syed Muzamil H Zaidi. PGY-3 Mentor: Dr S. Jayanth Samuel M.D., Ph.D VAMC Buffalo,NY
Heat Shock Proteins • A group of molecular Chaperones. • Chaperones are molecules mostly proteins with diverse functions especially protein folding. • While the polypeptide chain is being synthesized, the intermediary forms are stabilized by these proteins before they can assume their final conformation.
How are they induced? • Heat shock proteins are induced by various stresses including heat, Toxins,Radiation, Infection etc.
What do they do? • HSP are involved in: • Protein Synthesis. • Protein translocation. • Apoptosis. • Protein Degradation. • Signaling. • Antigen Presentation
How and where do they work? • HSP 70 - Cytosol, mitochondria,ER • HSP 90 - Cytosol ER • HSP 40 - Cytosol. • HSP 60 - Cytosol and mitocondiria • HSP 47 - ER
Clinical Implications: • In Cystic Fibrosis, mutation delays the dissociation of Cl channel protein from its chaperones so resulting in abnormal folding and loss of function.
Inflammatory Bowel Disease • Inducible Hsps have a significant role in gut inflammation in IBD. • Cytoprotective agents, protecting IECs from inflammation induced changes in function. • Immunomodulatory agents stimulating lymphocytes in target specific manner. Hu S, Ciancio MJ, Gastroenterology 2007;133:1893-1904
Tumor Immunity • Tumor cells release various Hsps. • These are immunogenic. • HSP peptide complexes are distinct from tumor to tumor. • Possibility of new treatments
Heat Responsive Protein - 12 • Homology with Hsp 90 and Hsp 70. • Both Hrp-12 mRNA and Protein increase with Stress. • Molecular weight is 12 KD.
Discovery • While the LDIF (Liver derived immunoinhibitory factor) was purified, an unexpected 12 kd protein was co purified. • Although LDIF is 28 kd protein, hrp was shown to purify with this column and later be reducible to 12 kd size, indicating a possible dimeric form. Tzung SP et al,Hepatology 1991; 14:888-894
Methodology • Purification: Livers from C57BI/6 mice were prepared by homogenization and delipidation and set to column chromatography. • The column was further purified by Reversed Phase Liquid chromatography. • The sample was concentrated and suspended in SDS-PAGE sample buffer and ran on a 15% Polyacrylamide gel. Sameul SJ et al,1997 Hepatology 25,1213-1222
Electrophoresis • Under reducing conditions (ME), most of Hrp12 is in 12 kd conformation • Under non reducing conditions, a minor 28 kd band was visualized. • Lane 1,Liver Cytosol; 2, Hrp Red;Hrp Non Red
Cloning & Sequencing • The 12 kd bands were excised and subjected individually to N terminal sequencing. • The determined amino acid sequence was used to design a pool of 128 oligonucleotides, each 20 bp in length. • These represented all possible nucleotide sequences for first 7 amino acids. • These were now applied to commercially available murine cDNA library. Sameul SJ et al,1997 Hepatology 25,1213-1222
Evaluation of cDNA • 1240 bp long. • Open Reading frame was 521bp long. • It codes for 173 amino acids with mol mass 18.5 kd. • It was variably phosphorylated and no bands were present in 12 kd range if reaction did not contain PKC and phosphatidyl serine.
Similarity • Search of amino acids and nucleotide sequences in Gene bank data identified two rat proteins: PSP and LKPS • 38-136 residues are identical. • With Hsp70/DnaK, Hrp has a significant homology too.
Comparison with PSP/LKPS & HSP • *…..Sites for phosphorylation by Protein kinase C • IIRKVIS……mitochondrial targeting signal • Hsp90-like sequence between L(84) and Y(138
Response to Heat • Hepa1-6 cells (murine hepatoma) in monolayers were exposed to heat and RNA was harvested at various stages. • There was an unexpected oscillatory pattern of mRNA expression as the cells were exposed to different temperatures. • The most consistent response was at 44.5O
Steady State levels of Hrp12 and Hsp90 mRNA normalized with Actin mRNA
Protein folding abnormalities • Implicated in Alzheimer’s. • Huntington’s Chorea. • Parkinson’s Disease. • Type 2 Diabetes. • Prion Disease
Tissue Distribution of Hrp12 • Major signal in Liver and Kidney on Northern Blots. • This detected the relatively lower presence of Hrp12 mRNA in Brain and lung. • A zooblot revealed that Hrp gene is highly conserved in all mammalian species.
Tissue specificity of HRP12 Northern blot of mouse tissues Western blot of mouse tissues Hrp12 Actin liv kid int hrt lun spl ske brn tes kid ske liv lun spl brn hrt
Cellular Distribution • Methodology: • Construction of bacterial expression vectors pGST-Hrp12 and pGST-C, its expression and preparation of antisera. • Preparation of mouse tissue extracts. • Western blots of these extracts. • Subcellular fractionation of Liver. • Immunohistochemistry • Immunofluorescence
Liver • IHC and IF showed intense staining in periportal region. • A coarsely granular staining in cytoplasm was the most consistent pattern. • Laser Confocal Microscopy of mouse Liver
Kidneys • Intense staining on the luminal border due to Alkaline Phosphatase. • Not removed by levamisole. • Most staining in PCT and DCT to a lesser extent.
What does it do? • The yeast-based 2 hybrid system is a way to identify protein-protein interactions. • Chimeric proteins are created by fusing the binding domain of an Transcription Activator to a bait protein (hrp12) and co-expressing this protein with a prey protein fused to the activation domain of the same Transcription Activator in yeast cells. • Interaction of the bait and prey proteins re-constitutes the Transcription Activator and a reporter gene is expressed
From Bench To Bedside! • TTR is a Homotetrameric protein secreted by the liver. • It binds retinol-binding protein and weakly to Thyroxine. • Much interest centers on how it multimerizes in the ER and how it is secreted. • 2 disorders of TTR tetramer stability are known: senile systemic amyloidosis (SSA) and familial amyloidotic polyneuropathy (FAP). • TTR can form amyloid in CSF (synthesized by the only other organ that makes it, i.e. choroid plexus, and has been implicated in depression and Alzheimer’s.
Acknowledgements • Dr Michael Sitrin M.D. • Valerie Anderson.