Biochemistry
Biochemistry. Metabolism. Cellular Metabolism. Metabolism. All the chemical reactions in the body; or acquiring and using energy Digestion: catabolism (breakdown) of nutrients in the GI tract for absorption ATP production for cellular energy Anabolism to build cellular structures

Biochemistry
E N D
Presentation Transcript
Biochemistry Metabolism
Metabolism • All the chemical reactions in the body; or acquiring and using energy • Digestion: catabolism (breakdown) of nutrients in the GI tract for absorption • ATP production for cellular energy • Anabolism to build cellular structures • Catabolism to recycle macromolecules • Movement
Digestion • Carbohydrates, lipids, and proteins are broken down into their simplest units by digestive enzymes • Carbohydrates to glucose by amylase, maltase, lactase, sucrase • Lipids to monoglycerides + fatty acids by lipase; transported in chylomicrons; changed in the cells to glycerol + fatty acids • Proteins to amino acids by pepsin, trypsin & others
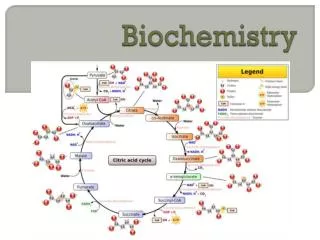
ATP Production • Glycolysis: breaking down glucose to produce a net of 2 ATP + pyruvate • Aerobic or anaerobic respiration • Anaerobic respiration when oxygen is lacking produces lactate (lactic acid) which is sent to the liver for processing • Aerobic respiration continues in the mitochondria with the citric acid cycle and electron transport chain
Important Coenzymes • Energy carriers: NAD+ and FAD bind hydrogens with high energy electrons and transport them to other reactions • NAD+ + 2H NADH + H+ (has energy) • FAD + 2H FADH2 (has energy) • Coenzyme A activates the acetyl group in pyruvate to acetyl CoA needed for the citric acid cycle
Glycolysis • 2 ATP are used to start the breakdown of 6-carbon glucose to two 3-carbon glyceraldehyde phosphates • Glyceraldehyde phosphate is converted to 4 ATP plus hydrogens that are given to NAD+
Metabolism of Pyruvate • With oxygen present, NADH and CoA react with pyruvate to form acetyl CoA • With a lack of oxygen, NADH is used to convert pyruvate to lactate
Mitochondria • Mitochondria, double-membrane organelles in the cell, converts pyrvate to acetyl CoA and then release energy from acetyl CoA to make 36 ATP • Cristae: folds in the inner membrane • Matrix: area inside the inner membrane where the citric acid cycle occurs • Intermembrane space: between the two membranes where H+ are concentrated • Inner membrane has protein enzymes for the electron transport chain
Citric Acid Cycle • Cyclic pathway of chemical reactions that releases energy to NADH and FADH2 and forms one GTP (converts to ATP) • Part one begins when a 2-carbon acetyl CoA combines with a 4-carbon oxaloacetate to form citrate (citric acid) • Part one removes 2 carbons • Part two converts the resulting 4-carbon compound back to oxaloacetate to begin another cycle
Electron Transport Chain • Enzymes use the energy in the high-energy electrons carried by NADH and FADH2 to transport H+ to the intermembrane space • Concentrated H+ pass quickly through a H+ channel associated with ATPase to produce ATP • H+ inthe matrix must combine with O2 to prevent a rapid drop in pH which would prevent further metaoblism
Metabolism • Carbohydrates, lipids, and proteins can be used for energy as well as for building cellular structures
Metabolism • Carbohydrates glucose for glycolysis • Extra glucose is stored as glycogen by the liver and muscle cells for later use • Lipids fatty acids and glycerol • Glycerol is converted in glyceraldehyde phosphate and enters the glycolysis pathway • Fatty acids are converted into acetyl CoA for the citric acid cycle • Proteins amino acids • Deamination removes the amine group • Deaminated molecules are converted into various reactants in the citric acid cycle
Metabolism • When the body uses fats and amino acids for energy, an excess of acetyl CoA can accumulate in the liver • Excessive acetyl CoA combines to form ketone bodies • Ketone bodies can be sent to the cells for a small amount of ATP production • Ketosis: when the ketone bodies are not completely metabolized • Leads to acidosis (low blood pH) which causes rapid, labored breathing