Carbon nanotubes
Carbon nanotubes. John, Sarah, Doug. Carbon nanotubes. Main interest: Prototypes for a one-dimensional quantum wire. Strength . Who found first nanotube?.

Carbon nanotubes
E N D
Presentation Transcript
Carbon nanotubes • John, Sarah, Doug
Carbon nanotubes Main interest: Prototypes for a one-dimensional quantum wire. Strength
Who found first nanotube? 1970: Morinobu Endo-- First carbon filaments of nanometer dimensions, as part of his PhD studies at the University of Orleans in France. He grew carbon fibers about 7 nm in diameter using a vapor-growth technique. Filaments were not recognized as nanotubes and were not studied. 1991:Sumio Iijima-- NEC Laboratory in Tsukuba-- used high-resolution transmission electron microscopy to observe carbon nanotubes.
Graphite • Hexagonal graphite: • Graphite has a structure containing layers of atoms arranged at the corners of contiguous hexagons. • (not to be confused with hexagonal close packed). • The ease with which layers slide against each other is consistent with the much larger distance between carbon atoms in different layers (335 pm) than between carbon atoms in the same layer (142 pm). • The lattice constant a = 246.6 pm • C=669 pm
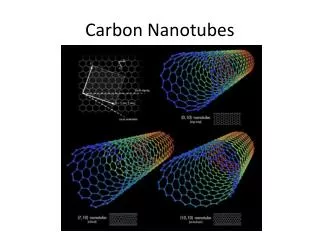
What is tube rolled out of ? An ideal nanotube can be thought of as a hexagonal network of carbon atoms that has been rolled up to make a cylinder. width: nanometer: "capped" with half of a fullerene molecule. length: microns
How to roll the nanotube ? • A carbon nanotube is based on a two-dimensional graphene sheet. • The chiral vector is defined on the hexagonal lattice as Ch = nâ1 + mâ2, • Chiral angle • Role/cap off
Different types of nanotubes (n, 0) or (0, m) and have a chiral angle of 0°, armchair nanotubes have (n, n) and a chiral angle of 30°, while chiral nanotubes have general (n, m) values and a chiral angle of between 0° and 30°.
What are the main properties? • Diameter of nanotube • Chiral angle • Both depend on n and m. • Diameter=length of chiral vector divided by 4 • (has to do with the capping)
Interesting thought from paper. Quote: “Since each unit cell of a nanotube contains a number of hexagons, each of which contains two carbon atoms, the unit cell of a nanotube contains many carbon atoms. If the unit cell of a nanotube is N times larger than that of a hexagon, the unit cell of the nanotube in reciprocal space is 1/N times smaller than that of a single hexagon. “ Let us break this down!
Interesting thought from paper. Then in reciprocal space: (pi/L) = (pi/a N) = (pi/a)(1/N) For k space
Dispersion relation in nanotubes 5,5 9,0 10,0
Some thoughts: E(kx, ky) Then because we have a cylinder: E(kxn , ky) Where Kxn = ((2*pi) /P) * n n=1,2,3 …(1-D bands)
How to make nanotubes? • Rice University group (1996)-- produce bundles of ordered single-wall nanotubes : • Prepared by the laser vaporization of a carbon target in a furnace at 1200 °C. • Cobalt-nickel catalyst helps the growth of the nanotubes, presumably because it prevents the ends from being "capped" during synthesis. • By using two laser pulses 50 ns apart, growth conditions can be maintained over a larger volume and for a longer time. This scheme provides more uniform vaporization and better control of the growth conditions. • Flowing argon gas sweeps the nanotubes from the furnace to a water-cooled copper collector just outside of the furnace. • Catherine Journet, Patrick Bernier and colleagues at the University of Montpellier in France: carbon-arc method to grow arrays of single-wall nanotubes.
Look like ? Scanning electron microscope: looks like a mat of carbon ropes Ropes are between 10 and 20 nm across and 100 µm long. Transmission electron microscope: each rope is found to consist of a bundle of single-wall carbon nanotubes aligned along a single direction. X-ray diffraction, which views many ropes at once, shows that the diameters of the single-wall nanotubes have a narrow distribution with a strong peak.