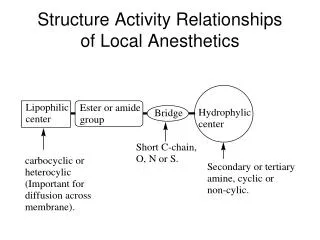
Structure-Activity Relationships
Structure-Activity Relationships. Drug Design. Barbiturates - . pharmacological and structural class. usually administered as salts. Modifications. Uses: sedative hypnotic antianxiety. An Antiischemic, Bradycardic Therapeutic Agent. bradycardia - slowed heartbeat.

Structure-Activity Relationships
E N D
Presentation Transcript
Structure-Activity Relationships Drug Design
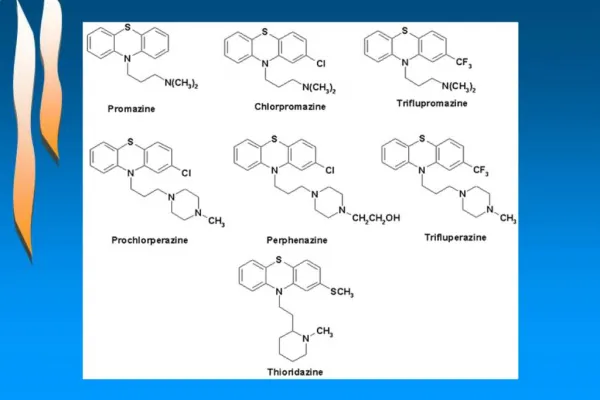
Barbiturates - pharmacological and structural class usually administered as salts Modifications • Uses: • sedative • hypnotic • antianxiety
An Antiischemic, Bradycardic Therapeutic Agent bradycardia - slowed heartbeat ischemia - condition in which the heart is deprived of oxygen and toxic metabolites cannot be removed. [Ca2+] very low in the cytosol. Ca2+-channels allow movement of calcium ions away from myosin to control muscle contraction. This drug would slow down the heart rate in order to decrease the oxygen demand. However, it is not desirable to lower the contractile force. Existing calcium-channel blocking drugs, while inducing bradycardia, also decrease the contractile force. This can lead to congestive heart failure.
Therapeutic goals - reduce heart rate, maintain contractile force, increase duration of action calcium-channel blocker
Cardiac Stimulant • Uses - • antiarrhythmic • prevent postmyocardial infarction • disadvantages include • I.V. administration (titration) • rapid biotransformation (t1/2 = 1.5-2 hours) • CNS toxicity Therapeutic goals - • oral administration • longer duration • separation of CNS toxicity from antiarrhythmia • patentable
Conclusions: • increase in lipophilicity leads to an increase in potency • increase in pKa leads to a decrease in toxicity • don’t discount toxic side effects
Lipinski’s Rule of 5 - a drug candidate will not be a good candidate for oral bioavailability if: • There are more than 5 H-bond donors. • The molecular weight is over 500. • The LogP is over 5 (LogP refers to the octanol-water partition coefficient, calculated based on MDL's QSAR program). • There are more than 10 H-bond acceptors. • Number of rotatable bonds >10. B.S.SF State Ph.D. Berkeley postdoc Cal Tech Christopher Lipinski (Pfizer) screened >2300 compounds to develop the four characteristics quickly adopted by pharmaceutical companies for early ADME E.B. Hersherg Award for Important Discoveries in Medicinally Active Substances(2004); 2004 ACS Division of Medicinal Chemistry Award
fragment-based scaffolding Fragments of Active Structures (FAST®) - SGX Pharmaceuticals • Use of library of small molecule (<300 D) • Crystallographic screening of shape-diverse mixtures of fragments to identify those bound to the protein target of interest. • Utilization of structurally accessible synthetic handles for virtual construction of “linear” elaborated libraries. • Computational analysis of these virtual libraries to identify those with favorable calculated binding free energies. • Linear and combinatorial library synthesis. • Biochemical analysis followed by selection of key analogs for analysis with protein:ligand crystal structures. • Iterative synthesis of additional compounds based on structural, novel chemical space, and synthetic reasons to optimize lead series.
fragment-based scaffolding SGX Pharmaceuticals Developed a library of small molecules (fragments) used to produce lead compounds. 1400 fragments - 140 shape-diverse pools
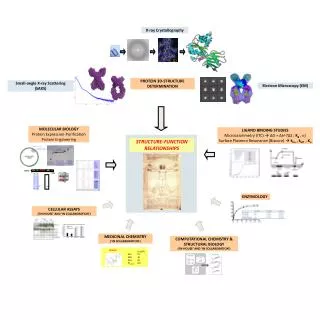
fragment-based scaffolding Targets kinases phosphatases nuclear receptors polymerases proteases active sites, allosteric sites, new(novel) sites Techniques X-ray crystallography (binding) Enthalpy Array (H of ligand binding) Nanocalorimetry SPR -plasmon resonance (kinetics of ligand binding)
Nano-calorimetry - enthalpy array Scripps PARC Institute enthalpy of ligand binding http://www.parc.com/research/projects/enthalpyarray/images/fig-01a-lg.jpg
Nano-calorimetry - enthalpy array http://www.parc.com/research/projects/enthalpyarray/images/fig-01a-lg.jpg
Plasmon Resonance sample http://employees.csbsju.edu/hjakubowski/classes/ch331/bind/olbindderveq.html
Computer-assisted analysis of carcinogenicity QSAR/QSPR (quantitative structure-activity relationship) (quantitative structure-property relationship) molecular descriptors physiochemical parameters topological geometry electron density
Drug X’ enantiomer of X Drug X D D C A A C B B E E F F Formulations, Durability, Optical Isomerism physical state physical properties chemical stability formulation compatibility receptor receptor A,F –hydrogen bond donors B,E– hydrogen bond acceptors C– hydrophobic group