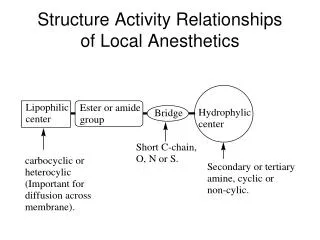
Quantitative Structure- Activity Relationships (QSAR)
Quantitative Structure- Activity Relationships (QSAR) Rationale for QSAR Studies In drug design, in vitro potency addresses only part of the need; a successful drug must also be able to reach its target in the body while still in its active form.

Quantitative Structure- Activity Relationships (QSAR)
E N D
Presentation Transcript
Rationale for QSAR Studies • In drug design, in vitro potency addresses only part of the need; a successful drug must also be able to reach its target in the body while still in its active form. • The in vivo activity of a substance is a composite of many factors, including the intrinsic reactivity of the drug, its solubility in water, its ability to pass the blood-brain barrier, its non- reactivity with non-target molecules that it encounters on its way to the target, and others.
Rationale for QSAR Studies... • A quantitative structure-activity relationship (QSAR) correlates measurable or calculable physical or molecular properties to some specific biological activity in terms of an equation. • Once a valid QSAR has been determined, it should be possible to predict the biological activity of related drug candidates before they are put through expensive and time-consuming biological testing. In some cases, only computed values need to be known to make an assessment.
History of QSAR • The first application of QSAR is attributed to Hansch (1969), who developed an equation that related biological activity to certain electronic characteristics and the hydrophobicity of a set of structures. log (1/C) = k1log P - k2(log P)2 + k3s + k4 for: C = minimum effective dose P = octanol - water partition coefficient s = Hammett substituent constant kx= constants derived from regression analysis
Hansch’s Approach • Log P is a measure of the drug’s hydrophobicity, which was selected as a measure of its ability to pass through cell membranes. • The log P (or log Po/w) value reflects the relative solubility of the drug in octanol (representing the lipid bilayer of a cell membrane) and water (the fluid within the cell and in blood). • Log P values may be measured experimentally or, more commonly, calculated.
Calculating Log P Log P = Log K (o/w) = Log ([X]octanol/[X]water) • most programs use a group additivity approach: 1 Aromatic ring 0.780 7 H’s on Carbon 1.589 1 C-Br bond -0.120 1 alkyl C 0.195 Sum = 2.924 = calc. log P • some use more complicated algorithms, including factors such as the dipole moment, molecular size and shape.
Hansch’s Approach... • The Hammett substituent constant (s) reflects the drug molecule’s intrinsic reactivity, related to electronic factors caused by aryl substituents. • In chemical reactions, aromatic ring substituents can alter the rate of reaction by up to 6 orders of magnitude! • For example, the rate of the reaction below is ~105 times slower when X = NO2 than when X = CH3
Hammett Equation • Hammett observed a linear free energy relationship between the log of the relative rate constants for ester hydrolysis and the log of the relative acid ionization (equilibrium) constants for a series of substituted benzoic esters & acids. log (kx/kH) = log (Kx/KH) = rs • He arbitrarily assigned r, the reaction constant, of the acid ionization of benzoic acid a value of 1.
Definition of Hammett r These sp values are obtained from the best fit line having a slope = 1
Hammett Plot • Aryl substituent constants (s) were determined by measuring the effect of a substituent on a reaction rate (or Keq). These are listed in tables, and are constant in widely different reactions. • Reaction constants (r) for other reactions may also be determined by comparison of the relative rates (or Keq) of two differently substituted reactants, using the substituent constants described above. • Some of these values (s and r) are listed on the following slide.
Hammett Rho & Sigma Values Reaction (Rho) Values r Substituent (Sigma) Values s (the electronic effect of the substituent; negative values are electron donating) p-NH2 -0.66 p-Cl 0.23 p-OCH3 -0.27 p-COCH3 0.50 p-CH3 -0.17 p-CN 0.66 m-CH3 -0.07 p-NO2 0.78
Molecular Properties in QSAR • Many other molecular properties have been incorporated into QSAR studies; some of these are measurable physical properties, such as: • density pKa • ionization energy boiling point • Hvaporization refractive index • molecular weight dipole moment (m) • Hhydration reduction potential • lipophilicity parameter p = log PX - log PH
Molecular Properties in QSAR • Other molecular properties (descriptors) that have been incorporated into QSAR studies include calculated properties, such as: • ovality surface area, molec. volume • HOMO energy LUMO energy • polarizability charges on individual atoms • molecular volume solvent accessible surface area • vdW surface area maximum + and - charge • molar refractivity hardness • hydration energy Taft’s steric parameter
QSAR Methodology • Often it is found that several descriptors are correlated; that is, they describe observables that are closely related, such as MW and boiling point in a homologous series. • Statistical analysis is used to determine which of the variables best describe (correlate with) the observed biological activity, and which are cross-correlated. The final QSAR involves only the most important 3 to 5 descriptors, eliminating those with high cross-correlation.
Limit to the # of Descriptors • The data set should contain at least 5 times as many compounds as descriptors in the QSAR. • The reason for this is that too few compounds relative to the number of descriptors will give a falsely high correlation: • 2 points exactly determine a line (2 comp’ds, 2 prop) • 3 points exactly determine a plane (etc., etc.) • A data set of drug candidates that is similar in size to the number of descriptors will give a high (and meaningless) correlation.
Statistical Analysis of Data • Multiple linear regression analysis can be accomplished using standard statistical software, typically incorporated into sophisticated (and expensive) drug design software packages, such as MSI’s Cerius2 (academic price, over $20K) • An inexpensive statistical analysis software StatMost (academic price, $39) works just fine. • To discover correlated variables and determine which descriptors correlate best, a partial least squares or principal component analysis is done.
Example of a QSAR Anti-adrenergic Activity and Physicochemical Properties of 3,4- disubstituted N,N-dimethyl-a-bromophenethylamines p =Lipophilicity parameter s+=Hammett Sigma+ (for benzylic cations) Es(meta) = Taft’s steric parameter
Example of a QSAR... Calc. Calc. m-X p-Yps+Es(meta)log (1/C)obslog (1/C)alog (1/C)b H H 0.00 0.00 1.24 7.46 7.82 7.88 F H 0.13 0.35 0.78 7.52 7.45 7.43 H F 0.15 -0.07 1.24 8.16 8.09 8.17 Cl H 0.76 0.40 0.27 8.16 8.11 8.05 Cl F 0.91 0.33 0.27 8.19 8.38 8.34 Br H 0.94 0.41 0.08 8.30 8.30 8.22 I H 1.15 0.36 -0.16 8.40 8.61 8.51 Me H 0.51 -0.07 0.00 8.46 8.51 8.36 Br F 1.09 0.34 0.08 8.57 8.57 8.51 H Cl 0.70 0.11 1.24 8.68 8.46 8.60 Me F 0.66 -0.14 0.00 8.82 8.78 8.65 H Br 1.02 0.15 1.24 8.89 8.77 8.94 Cl Cl 1.46 0.51 0.27 8.89 8.75 8.77 Br Cl 1.64 0.52 0.08 8.92 8.94 8.94 Me Cl 1.21 0.04 0.00 8.96 9.15 9.08 Cl Br 1.78 0.55 0.27 9.00 9.06 9.11 Me Br 1.53 0.08 0.00 9.22 9.46 9.43 H I 1.26 0.14 1.24 9.25 9.06 9.26 H Me 0.52 -0.31 1.24 9.30 8.87 8.98 Me Me 1.03 -0.38 0.00 9.30 9.56 9.47 Br Br 1.96 0.56 0.08 9.35 9.25 9.29 Br Me 1.46 0.10 0.08 9.52 9.35 9.33
Example of a QSAR... QSAR Equation a: (using 2 variables) log (1/C) = 1.151 p - 1.464 s+ + 7.817 (n = 22; r = 0.945) QSAR Equation b: (using 3 variables) log (1/C) = 1.259 p - 1.460 s+ + 0.208 Es(meta) + 7.619 (n = 22; r = 0.959)
Example of a QSAR... Calc. Calc. m-X p-Yps+Es(meta)log (1/C)obslog (1/C)alog (1/C)b H H 0.00 0.00 1.24 7.46 7.82 7.88 F H 0.13 0.35 0.78 7.52 7.45 7.43 H F 0.15 -0.07 1.24 8.16 8.09 8.17 Cl H 0.76 0.40 0.27 8.16 8.11 8.05 Cl F 0.91 0.33 0.27 8.19 8.38 8.34 Br H 0.94 0.41 0.08 8.30 8.30 8.22 I H 1.15 0.36 -0.16 8.40 8.61 8.51 Me H 0.51 -0.07 0.00 8.46 8.51 8.36 Br F 1.09 0.34 0.08 8.57 8.57 8.51 H Cl 0.70 0.11 1.24 8.68 8.46 8.60 Me F 0.66 -0.14 0.00 8.82 8.78 8.65 H Br 1.02 0.15 1.24 8.89 8.77 8.94 Cl Cl 1.46 0.51 0.27 8.89 8.75 8.77 Br Cl 1.64 0.52 0.08 8.92 8.94 8.94 Me Cl 1.21 0.04 0.00 8.96 9.15 9.08 Cl Br 1.78 0.55 0.27 9.00 9.06 9.11 Me Br 1.53 0.08 0.00 9.22 9.46 9.43 H I 1.26 0.14 1.24 9.25 9.06 9.26 H Me 0.52 -0.31 1.24 9.30 8.87 8.98 Me Me 1.03 -0.38 0.00 9.30 9.56 9.47 Br Br 1.96 0.56 0.08 9.35 9.25 9.29 Br Me 1.46 0.10 0.08 9.52 9.35 9.33
QSAR of Antifungal Neolignans • The PM3 semi-empirical method was employed to calculate a set of molecular properties (descriptors) of 18 neolignan compounds with activities against Epidermophyton floccosum, a most susceptible species of dermophytes. The correlation between biological activity and structural properties was obtained by using the multiple linear regression method. The QSAR showed not only statistical significance but also predictive ability. The significant molecular descriptors related to the compounds with antifungal activity were: hydration energy (HE) and the charge on C1' carbon atom (Q1'). The model obtained was applied to a set of 10 new compounds derived from neolignans; five of them presented promising biological activities against E. floccosum.
Descriptors Used • Log P: the values of this property were obtained from the hydrophobic parameters of the substituents; • superficial area (A) and molecular volume (V), log of the partition coefficient (Log P), hydration energy (HE): properties evaluated with the molecular modeling package HyperChem 5.0; • partial atomic charges (Qn) and bond orders (Ln) derived from the electrostatic potential; • energy of the HOMO (H) and LUMO (L) frontier orbitals; • hardness (h): obtained from the equation h =(ELUMO-EHOMO)/2; • Mulliken electronegativity (c): calculated from the equation c = -(EHOMO+ELUMO)/2; • other electronic properties were calculated: total energy (ET), heat of formation (DHf); ionization potential (IP), dipole moment (m) and polarizability (POL), whose values were obtained from the molecular orbital pprogram Ampac 5.0.
Antifungal QSAR Log 1/C = -2.85 - 0.38 HE - 1.45 Q1' F=29.63, R2=0.86, Q2=0.80, SEP=0. where: Fis the Fisher test for significance of the eq’n. R2 is the general correlation coefficient, Q2is the predictive capability, and SEP is the standard error of prediction. A.A.C. Pinheiro, R.S. Borges, L.S. Santos, C.N. Alves, Journal of Molecular Structure: THEOCHEM, Vol 672, pp 215-219 (2004).
3 Dimensional QSAR Methods • Important regions of bioactive molecules are “mapped” in 3D space, such that regions of hydrophobicity, hydrophilicity, H-bonding acceptor, H-bond donor, p-donor, etc. are rendered so that they overlap, and a general 3D pattern of the functionally significant regions of a drug are determined. • CoMFA (Comparative Molecular Field Analysis) is one such approach: testosterone
CoMFA of Testosterone Blue means electronegative groups enhance, red means Electn’g. gr’ps reduce binding Green means bulky groups enhance, yellow means they reduce binding