Section E DNA Replication
430 likes | 697 Views
Section E DNA Replication. E1-DNA Replication: An Overview E2-Bacterial DNA replication E3-The cell cycle E4-Eukaryotic DNA replication. E1: DNA Replication: An Overview Replicons, origins and termini. Prokaryotic genome : a single circular DNA = a single replicon;

Section E DNA Replication
E N D
Presentation Transcript
Section E DNA Replication E1-DNA Replication: An Overview E2-Bacterial DNA replication E3-The cell cycle E4-Eukaryotic DNA replication
E1: DNA Replication: An OverviewReplicons, origins and termini
Prokaryotic genome: a single circular DNA = a single replicon; Bidirectional replication of a circular bacterial replicon; All prokaryotic chromosomes and many bacteriophage and viral DNA molecules are circlular and comprise single replicons. There is a single termination site roughly 180o opposite the unique origin.
Linear viral DNA molecules usually have a single origin, replication details (see Section R). In all the cases,the origin is a complex regionwhere the initiation of DNA replication and the control of the growth cycle of the organism are regulated and co-ordinated..
Eukaryotic genome:multiple linear chromosomes& multiple replicons on each chromosome; Multipleeukaryotic repliconsand replication bubbles; The long, linear DNA molecules of eukaryotic chromosomes consist of mutiple regions, each with its own orgin.
A typical mammalian cell has 50000-100000 replicons with a size range of 40-200 kb. When replication forks from adjacent replication bubbles meet, they fuse to form the completely replicated DNA. No distinct termini are required.
Bidirectional replication of a circular bacterial replicon. Two replication forks proceed away from the orgin (O) towards the terminus (T). Daughter DNA is shown as a thick line. • Multiple eukaryotic replicons. Each origin is marked (●).
E1: DNA Replication An Overview semi-conservative mechanism Three models for DNA replication all obeyed base-pairing rules In each model, the stretches of original DNA are shown in blue and newly synthesized DNA in red. Watson and Crick’s original paper suggested that DNA replication is semiconservative.
Density Gradient Centrifugation The two strands of each DNA molecule stay together during extraction and centrifugation. Labeled (“heavy6”) DNA will separate from lighter DNA in the gradient formed by a cesium chloride solution.
E1: DNA Replication An Overview Semi-discontinuous Replication The Two Daughter Strands Form in different ways Okazaki fragments are hundreds of nucleotides long, with gaps between them.
Discovery of Okazaki fragments Evidence for semi-discontinuous replication • [3H] thymidine pulse-chase labeling experiment • Grow E. coli • Add [3H] thymidine in the medium for a few second spin down and break the cell to stop labeling analyze found a large fraction of nascent DNA (1000-2000 nt) = Okazaki fragments. • Grow the cell in regular medium then analyze the small fragments join into high molecular weight DNA= Ligation of the Okazaki fragments.
E1: DNA Replication An Overview RNA priming The first few nucleotides at the 5’-end of Okazaki fragments are ribonucleotides. Hence, DNA synthesis is primed by RNA that is then removed before fragments are joined. RNA priming is Crucial for high fidelity of replication.
Experimental systems • Two basic requires: • An Easy purified DNA: smaller and simpler bacteriophage and plasmid DNA molecules (fX174, 5 Kb). • It relies almost exclusively on cellular replication factors for its own replication.
Initiation Study system:the E. coliorigin locus oriCis cloned into plasmids to produce more easily studied minichromosomes which behave like E. coli chromosome.
1. OriC contains four 9 bp binding sites for the initiator protein DnaA. Synthesis of DnaA is coupled to growth rate so that initiation of replication is also coupled to growth rate. 2.DnaA forms a complex of 30-40 molecules, around which the oriC DNA becomes wrapped. This process facilitates melting of three 13 bp AT-rich repeat sequence which open to allow binding of DnaB protein.
3. DnaB is a helicase that use the energy of ATP hydrolysis to move into and melt the double-stranded DNA . 4.The single-stranded bubble created in this way is coated with single-stranded binding protein (SSb) to protect it from breakage and to prevent the DNA renaturing.
5. DNA primase:attaches to the DNA and synthesizes a short RNA primer to initiate synthesis of the leading strand of the first replication fork. 6. Primosome: a mobile complex including DnaB helicase and DNA primase synthesizes RNA primers every 1000-2000 nt on the lagging strand.
Re-initiationof bacterial replication at new origins before completion of the first round of replication. The theta (θ) mode of DNA replication in E.coli
Unwinding For replication to proceed away from the origin, DNA helicases must travel along the template strands to open the double helix for copying. In addition to DnaB, a second DNA helicase may bind to the other strand to assist unwinding. Binding of Ssb protein further promotes unwinding. Removal of helical turns at the replication fork leads to the introduction of additional turns in the rest of the molecular in the form of positive supercoiling.
This positive supercoiling must be relaxed continuously by the introduction of further negative supercoils by a type II topoisomerase called DNA gyrase.
Elongation • Both leading and lagging strand primers are elongated byDNA polymerase III holoenzyme: • Is a dimer complex, one half synthesizing the leading strand and the other lagging strand. • Having two polymerases in a single complex ensures that both strands are synthesized at the same rate • Both halves of the dimer contain an a subunit---polymerase • e-subunit---3’5’ proofreading exonuclease • b-subunit---clamp the polymerase to DNA
The remaining subunits in each half aredifferentand may allow the holoenzyme to synthesize short and long stretches of DNA on the lagging and leading strands, respectively.
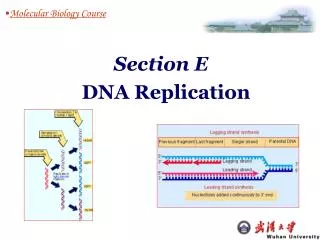
Other two enzymes during Elongation 1. Removal of RNA primer, and gap filling with DNA pol I 2. Ligation of Okazaki fragments are linked by DNA ligase. The laggig strand The lagging strand forms from left to right, while each Okazaki fragment forms from right to left- from its own 5’ end to its own 3’ end.
Replisome: in vivo, DNA polymerase holoenzyme dimer, primosome (helicase) are physically associated in a large complex to synthesize DNA at a rate of 900 bp/sec.
Termination and Segregation • Terminus: is in approximately 180o opposite oirC containing several terminator sites (ter). • Tus protein:ter binding protein, an inhibitor of the DnaB helicase and can arrest the movement of the forks by binding to terminus. • Topoisomerase IV: a type II DNA topoisomerase, which can unlink the interlinked daughter circles (genomes).
E3 The Cell cycle Cell Cycle Interphase 间期: G1 + S + G2 (G0) M phase (mitosis 有丝分裂):
G1 preparing for DNA replication (cell growth) S DNA replication G2 a short gap before mitosis M mitosis and cell division The Eukaryotic cell cycle A cell’s life history is made up of a short mitosis (purple) and a longer interphase (green). Interphase has three subphases (G1, S and G2) in cell that divide.
Cyclin-Dependent Kinase and Cyclin Trigger Decisions in the cell cycle A human cell makes the “decision” to enter the cell cycle during G1 when cyclin D binds to a cyclin-dependent kinase (Cdk4). There are four such cyclin-Cdk controls during the typical cell cycle in humans. Entry into the S-phase: Cyclins Cyclin-dependent protein kinases (CDKs) Signaling
E2: Eukaryotic DNA replication • Experimental system • Origins and initiation • Replication forks • Nuclear matrix • Telomere replication
experimental systems • Two basic requires: • An Easy purified DNA (genome) • Possess all the proteins and other factors for its complete replications
Small animal virus(simian virus 40, 5 kb) is an excellent model for a mammalian replication fork (for elongation but not for initiation). 2. Yeast(Saccharomyces cerevisiae): 1.4 X 107 bp in 16 chromosomes, only 400 replicons, much simpler than mammalian system and can be served as a model system. 3. Cell-free extractprepared from the eggs of the African clawed frog, Xenopus laevis contains high concentration of replication proteins and can support in vitro replication.
Origins and Initiation • 1. Clusters (tendem arrays) of about 20-50 replicons initiate simultaneously at defined times throughout S-phase. • Early S-phase: predominantly euchromatin replication. • Late S-phase: mainly heterochromatin replication. • Centromeric and telomeric DNA replicates last. • 2. Can Only initiate once per cell cycle • Licensing factor (A protein): • Is absolutely required for initiation and inactivated after use. • Can only gain access to nucleus when the nuclear envelope dissolves at mitosis.
3. origins Yeast replication origins (ARS- autonomously replicating sequences,allows the prokaryotic plasmids containing ARS to replicate in yeast). Minimal sequence of ARS: 11 bp [A/T]TTTAT[A/G]TTT[A/T] (TATA box) Additional copies of the above sequence is required for optimal efficiency. ORC (origin recognition complex) binds to ARS, and enable open the DNA for replication when activated by CDKs.
Replication fork 1-Unwinding DNA from parental nucleosomes at the replication forks: the movement of the forks to about 50 bp/sec, >1 DNA helicases and RP-A (a single-strand binding protein) 2-New nucleosomes are assembled to DNA from a mixture of old and newly synthesized histones after the fork has passed.
2, Elongation: three different DNA polymerases are involved. • DNA pol a: possess primase activity and can synthesize RNA primers for the leading strands and each lagging strand fragments. Continues elongation with DNA but is quickly replaced by the DNA polymerase-d (delta). • DNA pol d: replaces DNA pol a on the leading strand and probably also on the lagging strand and then continue to synthesize long DNA • DNA pol e (epsilon): is less clear, but it may simply complete the lagging strand fragments after primer removal. .
Nuclear Matrix Nuclear matrix is a scaffold of insoluble protein fibers which acts as an organizational frameworkfor nuclear processing, including DNA replication. Huge replication factories containing all the enzymes and DNA associated with the replication forks of all replicons within a cluster are immobilized on the matrix, and the DNA moves through these sites as it replicates. BUdR labeling of DNA Visualizing by immunoflurescence using BUdR antiboby Replication factories in a Eukaryotic nucleus
Telomere replication The ends of linear chromosomes cannot be fully replicated by semi-discontinuous replicationas there is no DNA to elongate to replace the RNA removed from the 5’-end of the lagging strand. Thus, genetic information could be lost from the DNA.
To overcome this, the ends of eukaryotic chromosomes (telomeres) consist of hundreds of copies of a simple, noninformational repeat sequence(e.g. TTAGGG in humans) with the 3’-end overhanging the 5’-end (Fig. 3). The enzyme telomerase contains a short RNA molecule, part of whose sequence is complementary to this repeat. This RNA acts as a template for the addition of these repeats to the 3’-overhang by repeated cycles of elongation and translocation.