Non-Biodegradable
The Advances and Challenges in Ocular Drug Delivery via Implantation Techniques. Silicone cup containing drug. EFFECT OF ENVIRONMENT AND CLOZAPINE ON BASAL AND STIMULATED MEDIAL PREFRONTAL GABA RELEASE IN TWO RAT MODELS OF SCHIZOPHRENIA. Top View. Release orifice. 3mm.

Non-Biodegradable
E N D
Presentation Transcript
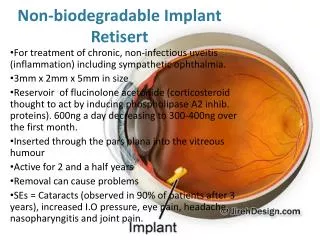
The Advances and Challenges in Ocular Drug Delivery via Implantation Techniques Silicone cup containing drug EFFECT OF ENVIRONMENT AND CLOZAPINE ON BASAL AND STIMULATED MEDIAL PREFRONTAL GABA RELEASE IN TWO RAT MODELS OF SCHIZOPHRENIA Top View Release orifice 3mm Adedoyin Awodele, Faye Carrington, AlannaCavanagh, Constance Gaya Cremers, Sarah Kileen, Melissa MacPherson.. Pharmacology, UCD School of Biomolecular and Biomedical Science, University College Dublin, Ireland. Side View PVA structure tab Introduction 2mm Ocular drug barriers: Why Implants? Diseases of the Eye 5mm • Age-related Macular Degeneration (AMD) : Damage to retina by accumulation of drusen can cause loss of central vision. • Provide sustained drug delivery to the posterioror anterior segment of the eye. • Localised drug delivery, therefore reduced dose. • Can be applied to various ocular layer depending on disease: subconjuctival/intravitreal/intrasceral. • Implants reduce frequency of administration • and risk of side effects. • Minimise the importance of patient compliance. • Drug can be removed should toxicity occur. • Diabetic Retinopathyis a type of retinal damage that can lead to blindness caused by microvascular changes due to diabetes mellitus. • Diabetic Macular Edema (DME) is the most common cause of visual loss and is characterised by accumulation of extracellular fluid in the macular which occurs after the break down of the blood-retinal barrier due to dilated hyper-capillaries and micro-aneurysms.. Above: Insertion of Retisert What are they? • Glaucomais caused by damage to the optic nerve by loss of retinal ganglion cells and increased fluid pressure in the eye. • .Usually made of polymers that release a drug over a prolonged period of time. There are two types: • Retinitis Pigmentosais a progressive retinal dystrophy which affects the photoreceptors and causes loss of peripheral vision and eventually central vision. • CMV Retinitisis a chronic, infection of the retina caused by the cytomegalovirus. Predominantly affects immunosuppressed individuals and estimated to affect 15-40% of AIDS patients. • Uveitis is the swelling and irritation of the uvea (anterior segment) and can be caused by autoimmune disorders, infection or exposure to toxins. Implants: The way forward? • Retisert • Vitrasert • FDA approved in 1996, Vitrasert was first non-biodegradable, intravitreal implant. • Used for the delivery of the anti-viral drug, ganciclovir to treat AIDs-related Cytomegalovirus (CMV) Retinitis. • Ganciclovir is a synthetic analogue of the nucleoside 2-deoxyguanosine, which causes chain termination, preventing replication. • Each implant holds 4.5-5mg of the prodrug which is released at a rate of approx. 1-1.5µg/hr over 5-8 months. • The 4mm device consists of a compressed drug pellet which is coated in the permeable, water-soluble polymer; PVA. This is completely surrounded, except at its top surface, with the impermeable polymer; EVA. The entire assembly is then covered by a 2nd layer of PVA. • For treatment of chronic, non-infectious uveitis (inflammation) including sympathetic ophthalmia. • 3mm x 2mm x 5mm in size • Reservoir of fluocinoloneacetonide (corticosteroid thought to act by inducing phospholipase A2 inhib. proteins). 600ng a day decreasing to 300-400ng over the first month. • Inserted through the pars plana into the vitreous humour • Active for 2 and a half years • Removal can cause problems • SEs = Cataracts (observed in 90% of patients after 3 years), increased I.O pressure, eye pain, headache, nasopharyngitis and joint pain. Side Effects and Complications of Implants Structure Vitreous Haemorrhage Endophthalimitis • 0.59mg tablet is held in a silicone elastomer cup. • The release orifice is separated from the drug • by a PVA membrane. • The structure is held together with silicone glue Non-Biodegradable Pros: In addition, to reduced systemic S/Es, increased efficacy and reduced dosing frequency, studies show treatment with vitrasert significantly slows down the median time to disease progression in comparison to I.V. ganciclovir therapy. Con: Increased risk of developing contralateral eye retinitis and systemic CMV. Cystoid Macular Oedema Retinal Detachment Advances in Implants/ Developments • Surgical Implantation • The implant is inserted by making a 5-6mm scleral incision into the pars plana. It is then fixed into place using sutures. The wound is closed and a saline solution is injected to restore normal ocular pressure. • Most patients experience blurred vision which usually clears between 2-4 weeks after surgery. • Injectable Implants: • Iluvien is a non-biodegradable implant that was recently approved as a treatment for DME. It is in phase II trials for the treatment of wet and dry AMD and RVO and its active ingredient is fluocinoloneacetonide. It can be delivered intravitreally using a 25 gauge needle, this is a mninimally invasive procedure as there is no need for a suture and is easier to deliver than retisert due to its smaller size. Implants vs. Other Deliveries Biodegradable Ozurdex Ozurdex is a biodegradable implant that contains demaxethasone. It is an intravitreal implant that delivers a sustained release of demaxaethasone (700ml) to the retina and vitreous humour. Ozurdex can improve visual acuity and macular thickness. It is used to treat macular edema, retinal vein occlusion and non-infectious uveitis (posterior). • Encapsulated Cell Technology (ECT) • ECT has been used in non-biodegradable implants and is a cell-based delivery system whereby implants can be used to deliver genetically modified cells in a hollow tube made from semi-permeable membrane with both ends of the polymer sealed. Ciliaryneurotrophic factor (CNTF) has been proved to protect the retina against degeneration. An ETC implant containing NTC-201 cells which were human retinal pigment epithelial cells that were modified to over produce CNTF were shown to significantly reduce retinal degeneration in animal models. Future Perspectives Ozurdex is made using a solid biodegradable polymer. This polymer is composed of an apolylactic acid-co-glycolic acid (PLGA) matrix. This dissolves completely in vivo. The products of this are lactic acid and glycolic acid, which are converted into Carbon dioxide and Water. Above : Implant on day 3 (A) and day 180 (B) References