Understanding Oxidation and Combustion in Arson Investigations
Explaining the relationship between oxidation and combustion in arson, including chemical processes, activation energy, and fire behavior. Learn about the chemistry of fire and common accelerants used, and understand the Fire Tetrahedron model.

Understanding Oxidation and Combustion in Arson Investigations
E N D
Presentation Transcript
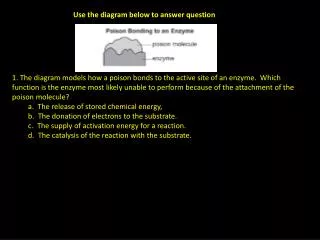
Answer Q Below • Explain the terms oxidation reaction and combustion reaction. How are these terms related?
Arson • Arson is defined as purposely setting fire to a house, building or other property • Arson is the second leading cause of death by fire in the U.S. As estimated 500 Americans died in arson-related fires
Chemistry of Fire • Chemically, fire is a type of oxidation, which is the combination of oxygen with other substances to produce new substances. • To start fire, the minimum temperature needed to spontaneously ignite fuel, known as ignition temperature, must be reached. • The heat evolved when a substance burns is known as heat of combustion. • An additional factor, besides the liberation of energy, needed to explain fire is the rate or speed at which the oxidation reaction takes place.
The Chemistry of Fire • A fuel will achieve a reaction rate with oxygen sufficient to produce a flame only when it is in the gaseous state. • A liquid burns when the temperature is high enough to vaporize it (flash point), while a solid must be hot enough to decompose into gaseous products (pyrolysis). • Glowing combustion or smoldering is burning at the fuel-air interface, such as a cigarette. • Spontaneous combustion, which is rare, is the result of a natural heat-producing process in poorly ventilated containers or areas.
Oxidation Review • oxidation the complete or partial loss of electrons or the gain of oxygen • OIL RIG or LEO says GER • 2Na + Cl2 2NaCl • CH4 + 2O2 2 H2O + CO2
Combustion • oxidation reactions that involve oxygen and produce flames • these reactions release energy in the form of heat and light
Combustion reactions • Subclass of oxidation-reduction reactions • Organic compounds react with oxygen to form carbon dioxide and water vapor. • Other compounds also formed: carbon monoxide, carbon (soot), poisonous gases (nitrogen oxides and sulfur dioxide) and small hydrocarbons • Color of flame and presence or absence of smoke are indicators of how efficient the combustion is.
Why does fire need heat? • Liquid and solid fuels need to vaporize • Pyrolysis – when fuels decompose to form gaseous products that are combustible • Activation energy – combustion is a chemical reaction. Bonds are broken and products are formed. Energy is needed to break bonds.
Activation energy • Energy needed to initiate a reaction • Usually a spark or flame • Flashover- occurs without a spark • Heat from fire rises to ceiling and spreads throughout room. • Temperature of furniture increases until auto-ignition temperature is reached • Fire spreads from point of origin to entire room.
The Fire Tetrahedronmodel used to describe how a fire starts and keeps burning
HEAT OXYGEN Fuel can be any combustible material in any state of matter - solid, liquid, or gas. Most solids and liquids become a vapor or gas before they will burn. Examples: CLOTHING FURNITURE CURTAINS FLAMMABLE LIQUIDS FUEL
HEAT OXYGEN The air we breathe is about 21% oxygen. Fire requires an atmosphere with at least 16% oxygen. FUEL
Remember: Fuel + Oxygen + Heat = Fire Heat is the energy necessary to increase the temperature of the fuel to a point where sufficient vapors are given off for ignition to occur. Examples: STOVES HEATING APPLIANCES FIREPLACES DAMAGED WIRING FUEL HEAT OXYGEN
FUEL HEAT OXYGEN • Chain reaction • The oxidation reaction (burning) must produce enough heat to maintain burning • The fuel mass must be vaporized and drawn up into the flame
Flash point- The temperature at which a particular flammable liquid gives off vapors (vaporizes) and therefore can ignite. Theignition temperatureis the temperature required for a liquid to continue to emit vapors that can sustain
Accelerants are any liquid, solid or gaseous material that will sustain or enhance flammability. Liquid materials are commonly used because of ease of ignition and familiarity of use. Accelerants are nearly exclusively derived from hydrocarbons. Straight chain hydrocarbons are the backbone of the oil industry. Hydrocarbons are molecules made up of the elements hydrogen and carbon. Octane is a term familiar to all. It consists of a hydrocarbon having 8 carbons. Accelerants
Accelerants • Examples: • Gasoline • Kerosene • Diesel • Lighter fluids • Charcoal starters • Automobile additives • Camping fuels
Ignition temperature. • Combustion will continue until: • Fuels are consumed. • Oxidizing agent has been removed. • Fuels are cooled below their ignition temperature. • Flames are chemically retarded. • Transfer of heat. : • Conduction. • Convection. • Radiation. • Direct flame contact.
Point of Origin – Burn patterns and other damage can help determine the point of origin, or the location where the fire started. First the point of origin is determined: • Where the fires starts • Usually contains the most damage. Once point of origin is determined, the cause of the fire is determined.
Interior Examination. • Work backward in relation to fire travel and from least to most damage. • In accidental fires, floor damage is limited in respect to the ceiling damage. • “V” patterns may help locate • Point of Origin • Defined as where the fire originated. • Cause of fire may be near • Fire usually burns longer • If accelerants or ignition devices used, they may be present • Multiple points MAY indicate arson. • “V” patterns usually point to • Extensive ceiling damages may be present above
Fire Clues • V-Patterns - Fire burns up, in a V-shaped pattern, so a fire that starts at an outlet against a wall leaves a char pattern that points to the origin. • A very narrow V-shape might indicate a fire that was hotter than normal, such as one helped along by an accelerant. • A wide V-shape might indicate a fire that was slow burning. • A U-shape could indicate that there was a "pool of origin" rather than a point of origin, such as might be caused by, say, a puddle of gasoline.
Char Patterns • Created by very hot fires that burn very quickly and move fast along its path, so that there can be sharp lines between what is burned and what isn't. • A char pattern on a door would help an investigator determine which side of the door the fire was on. • A char pattern on the floor would help investigators determine the use of an accelerant and its path.