Heat, Temperature, and Expansion
310 likes | 1.12k Views
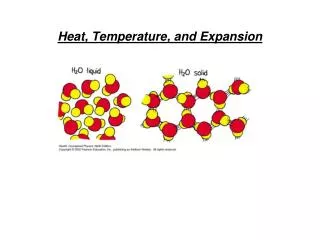
Heat, Temperature, and Expansion. Chapter 15. Temperature Heat Specific Heat Capacity Thermal Expansion. Heat &Temperature. Related to our concept of “hot” and “cold”, i.e. temperature is based on our sense of touch. It is also related to the kinetic energy of molecules of a material

Heat, Temperature, and Expansion
E N D
Presentation Transcript
Chapter 15 • Temperature • Heat • Specific Heat Capacity • Thermal Expansion
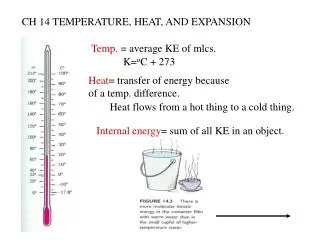
Heat &Temperature • Related to our concept of “hot” and “cold”, i.e. temperature is based on our sense of touch. • It is also related to the kinetic energy of molecules of a material • Temperature and Heat are fundamentally macroscopic concepts • Heat is energy in transit - measured in joules or calories. • Temperature is energy per molecule measured in degrees.
Temperature • Temperature is a measure of “hotness” or “coldness” on some scale. • The question is: What scale do we use? • We have to come up with a measuring device that measures some physicalproperty that varies with hotness or coldness. • The scale must be able to assign numbers to various states of hotness and coldness.
Temperature ScalesThe Celsius Scale • How does one create a scale? • It comes from experiment Divide this distance into 100 even parts = 1 oC intervals on the Celsius “scale” level of mercury when placed in boiling water -call this 100 OC observed level of mercury when placed in freezing pure water, call this 0oC Mercury, let’s say
The Fahrenheit Scale This scale was introduced by Gabriel Daniel Fahrenheit in 1720 who lived 1686-1736. The scale also uses two fixed points but instead of ice water and boiling water, he used the lowest temperature he could achieve at the time and assigned it a value of 32 and assigned the value of 96 to the reading of the scale when his thermometer was placed under a human armpit (or in the mouth).
Kelvin Scale • One can plot pressure P verses temperature oC P extrapolated data experimental data, data is linear 0 T (oC) - 273.15 oC a theoretical temperature at which the gas pressure would be zero.
Kelvin Scale • The theoretical temperature of -273.15 oC turns out to be the same for many different gasses. • -273.15 oC is not observed experimentally. • We use this extrapolated zero-pressure temperature as a basis of a newtemperature scale with it’s zero at this theoretical temperature. Named after Lord Kelvin 1824-1907
Kelvin Scale • The units are the same SIZE as those of the Celsius scale but the zero is shifted so that 0 K = - 273.15 oC • Tk = Tc + 273.15 • Room temperature of 20oC = about 293 K
TemperatureConversions • Useful formulas: Tk = Tc + 273.15 Tf = (9/5) Tc + 32o Tc = (5/9)(Tf - 32)where Tf is in Fahrenheit • This Kelvin scale is called an absolute temperature scale, and its zero point is called absolute zero. Most physics/chemistry formulas use kelvins.
Heat • Put spoon into coffee • spoon warms up and coffee cools down as they reach thermal equilibrium • We say energy has been transferred from one substance to another. • Energy transfers that occur only because of a temperature difference T is called heat flow or heat transfer and such energy transferred in this way is called HEAT.
Concept of Heat • Heat refers to a transfer of energy from one body to another • Heat DOES NOT refer to the amount of energy “contained” in a body or system. Heat always flows from the hotter object to the cooler object independent of the amount of “heat energy” each object contains
Specific Heat • Different substances have different capacities for storing internal energy • The specific heat capacity of any substance is defined as the quantity of heat required to change the temperature of a unit mass of the substance by 1 degree • Consider it as “thermal inertia” • The more heat energy it takes to raise a substances temperature, the more heat energy is given up by the substance as it cools down!!!
Thermal expansion of Materials • Suppose you have a rod with some initial length Lo at some initial temperature To. When the temperature changes by T, the length will change by an amount L. • Experiment shows that L is directly proportional to T provided T is less than about 100 oC. L Lo
Thermal expansion of Materials • Bimetallic strips • Example – thermostats • Expansion joints in roads, sidewalks, etc.
Water • What was the temperature at the bottom of Lake Michigan on October 31 1911?
Water • What was the temperature at the bottom of Lake Michigan on October 31 1911? – 4o C • Water is most dense at 4o C – most materials do not have this property! • Why does a lake freeze on top?
Class Problem • Touch the inside of a 220°C hot oven and burn yourself. But when the 1800°C white hot sparks form a 4th-of-July-type sparkler hit your skin, you're okay. Why?
Class Problem • Touch the inside of a 220°C hot oven and burn yourself. But when the 1800°C white hot sparks form a 4th-of-July-type sparkler hit your skin, you're okay. Why? • Temperature is proportional to energy per molecule. How much energy depends on how many molecules. When you touch the inside surface of the oven, you're making contact with many, many molecules, and the flow of energy is a painful experience. Although the energy per molecule is greater in the sparks of the firework, you make contact with only a relatively few molecules when a spark lands on you. The corresponding low energy transfer borders on your threshold of feeling.
Class Problem • When the temperature of a metal ring increases, does the hole become larger, smaller, or stay the same?
Class Problem • When the temperature of a metal ring increases, does the hole become larger, smaller, or stay the same? • When the temperature increases, the metal expands—in all directions. It gets thicker; its inner as well as its outer diameter increases; every part of it increases by the same proportion. To better see this, pretend that the ring is cut in four pieces before being heated. When heated they all expand. Can you see when they are reassembled that the hole is larger?Test this yourself the next time you can't open the metal lid on a jar. Heat the lid by placing it on a hot stove or under hot water so that its temperature momentarily increases more than the glass jar. Its inner circumference will increase and you'll easily unscrew the lid.
Class Problems 1. Heat energy travels from an object with a high A) internal energy to an object with a lower internal energy. B) temperature to an object with a lower temperature. C) Both of these, for they say essentially say the same thing. D) None of the above choices are true. 2. Which of the following normally warms up fastest when heat is applied? A) glass B) water C) wood D) iron E) All of the above choices are equally true.
Class Problems 3. Compared to a giant iceberg, a hot cup of coffee has A) higher temperature, but less internal energy. B) more internal energy and higher temperature. C) a greater specific heat and more internal energy. D) none of these 4. When water at 4 degrees C is heated it expands. When water at 4 degrees C is cooled, it A) expands. B) contracts. C) neither contracts nor expands.
Class Problems • 5. Aluminum has a specific heat capacity more than twice that of copper. Place equal masses of aluminum and copper wire in a flame and the one to undergo the fastest increase in temperature will be • A) Copper • B) Aluminum • C) both the same • 6. Some molecules are able to absorb large amounts of energy in the form of internal vibrations and rotations. Materials composed of such molecules have • A) high specific heats. • B) low specific heats. • C) none of the above