Estimating Persistence and Degradation Kinetics of Metabolites in Environmental Fate Studies
This document outlines the regulatory framework for evaluating the persistence and degradation kinetics of metabolites in the context of EU pesticide registration. It emphasizes the importance of exposure analysis and the establishment of kinetic endpoints as triggers for further studies. The formulation of kinetic models, particularly the Simple First Order (SFO) model and other bi-phasic models, are discussed to derive half-lives and degradation rates. The aim is to enhance understanding of metabolite behavior within various environmental compartments during risk assessments.

Estimating Persistence and Degradation Kinetics of Metabolites in Environmental Fate Studies
E N D
Presentation Transcript
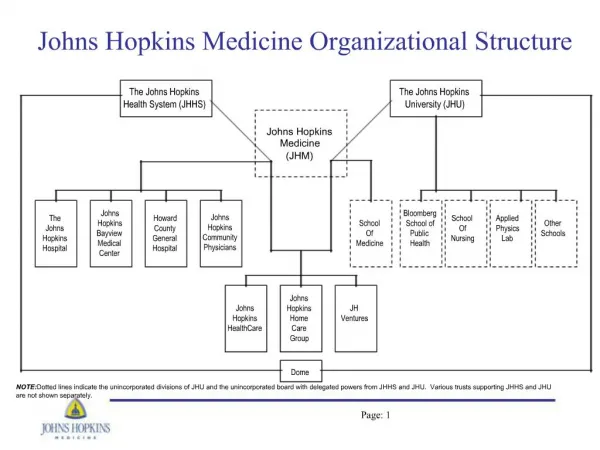
Theory Metabolites Karin Aden (BVL, Germany) FOCUS Work Group on Degradation Kinetics Estimating Persistence and Degradation Kinetics from Environmental Fate Studies in EU Registration Brussels, 26-27 January 2005 page 1
Introduction Regulatory Background - Triggers established in Annex VI of Directive 91/414/EEC must be applied to relevant metabolites - The assessment of the relevancy of a metabolite normally involves performing an exposure analysis (soil, groundwater, water-sediment-systems) - Kinetic endpoints are needed as triggers for subsequent studies for relevant metabolites, and for the modelling of the metabolites in the different environmental compartments - For metabolites applied as test substance, degradation kinetics should be derived following recommendations for parent (treated as parent substance) page 2
Parent Metabolite Sink kP->Met kMet->Sink kP->Sink Introduction Formulation of Kinetic Models Compartment Model: Equation SFO-Model: Data points are available and can be used for parameter estimation. Data points are not available OR if they are, with high uncertainty page 3
Metabolite Half-life: 23 d • SFO model 60 % ffMet=0.6 100 • 80 • Parent Half-life: 35 d 100 % • 60 • 40 • • • • • • • • 20 • 40 % 1-ffMet=0.4 Sink 0 0 20 40 60 80 100 Introduction Formation Fraction - Rate Constant - Overall Degradation Rate I • Metabolite formation fraction Maximum observed! • Sum of formation fractions started from one substance = 1 page 4
Introduction Formation Fraction - Rate Constant - Overall Degradation Rate II Formulation with rate constants: Example (SFO model): Parent: overall degradation rate kP: 0.02 d-1 (Half-life= 35 d) rate constant kPMet: 0.6 * kP = 0.012 d-1 formation fraction ffMet rate constant kPSink 0.4 * kP = 0.008 d-1 formation fraction 1-ffMet Metabolite: overall degradation rate kMet: 0.03 d-1 (Half-life= 23 d) Formulation with formation fractions: page 5
Introduction Formation - Plateau - Decline 100 Day 25: 1.2 =1.2 80 60 Day 70: 0.05 <0.67 40 20 Day 5: 4.9 >0.6 0 0 20 40 60 80 100 Formation phase kP*P > kMet*Met Plateau/Peak kP*P = kMet*Met Decline phase kP*P < kMet*Met SFO model Parent —100 %—> Metabolite page 6
max. amount: 60 % (25 d) • • 100 100 • 80 80 • • 60 60 • • • • • 40 40 • • • • • 20 20 • • • • • • 0 0 0 20 40 60 80 100 0 20 40 60 80 100 Which metabolite degraded faster? (SFO model, Parent —100 %—> Metabolite) Introduction Comparison of two Parent-Metabolite Systems Metabolite Half-life: 34 d Parent Half-life: 10 d Metabolite Half-life: 34 d Parent Half-life: 50 d Example 1 Example 2 max. amount: 30 % (59 d) • • • • • • • page 7
Types of Kinetic Models for Metabolites I • SFO model (Simple First Order) • Robust model, because of limited number of parameters (initial amount and rate constant for parent, formation fraction and rate constant for each metabolite). SFO is implemented in simulation models. The half-life calculation is simple. • Bi-phasic models • Hockey-stick model - should not be used! • Model with its single breakpoint time is not conceptually correct for a metabolite. Due to its continuous formation, deviations from SFO for a metabolite will appear to be gradual and smoothed. Parameter are often uncertain. • bi-exponentialDFOP model (Double-First-Order in Parallel) • DT50 values cannot be directly calculated from the model parameters although these trigger values can be derived using an iterative method. page 8
Types of Kinetic Models for Metabolites II • Bi-phasic models (cont.) • FOMC model (First Order Multi Compartment/Gustafson&Holden) • 1st Choice • FOMC can be easily implemented for metabolites with a single differential equation. It has only one additional parameter compared to the SFO model. The DT50 calculation of is simple. • FOMC model cannot be implemented in complex SW- and GW-models not valid for the determination of modelling endpoints, except PECsoil. • Exception: FOMC DT90 values of terminal metabolites, can be used as conservative estimate of the SFO Half-life by dividing the FOMC DT90 by 3.32. • This approach is only valid for terminal metabolites. Otherwise it would affect the kinetics of formation of metabolites further down in the degradation pathway! page 9
Types of Kinetic Models for Metabolites III DFOP-model: FMOC (Gustafson&Holden): page 10
Metabolite Endpoints Definition Distinction needs to be made between: 1.) Kinetics endpoints for metabolites used as triggers for higher tier experiments (“Trigger Endpoints”) and 2.) Kinetics endpoints used for modelling (“Modelling Endpoints“) page 11
Metabolite Endpoints Trigger Endpoints • Trigger Endpoints: Degradation/Dissipation DT50, DT90 • Derived by best-fit kinetics - unless deviations from SFO kinetics can be attributed to experimental artefacts • Trigger DegT50 and DegT90 values can be calculated from the estimated degradation rate of the metabolite using the equation corresponding to the best-fit kinetic model (consideration of the degradation only) • A conservative estimate of the trigger DegT50 and DegT90 values can be obtained by estimating the disappearance of the metabolite from its observed maximum, by fitting the decline curve (=consideration of the formation) page 12
100 Fit of the metabolite decline curve (SFO) 100 80 80 60 60 40 40 20 20 0 0 0 20 40 60 80 100 0 20 40 60 80 100 Metabolite Endpoints Trigger Endpoints - Example Time after application Time after maximum observed Half-life Parent: - Decline Metabolite (DissipationT50): Half-life: 114 d (0.006 0.0008) Fit of parent - metabolite system (both SFO) Half-life Parent: 13 d Degradation Metabolite: Half-life: 71 d (0.010 0.002) page 13
Metabolite Endpoints Modelling Endpoints • The required Modelling Endpoints for an individual metabolite are kinetic parameters and type of kinetic model used: • Formation rate parameters • • degradation rate parameters from precursor(s) • • formation fraction(s) • + • Degradation rate parameters • Usually SFO is used for modelling! page 14
Parent Metabolite Sink Main recommendations Pathway • Pathway • Conceptual model must reflect actual degradation or dissipation pathway • Flows to sink are initially included for formation of other metabolites (identified or not), bound residues and CO2 page 15
Metabolite Metabolite Parent Parent Sink Parent initial amount is not described properly (too low) Main recommendations Pathway - Example: Use of sink Half-life Parent: 6 d Half-life Metabolite: 16 d Formation fraction ffMet: 1 (fixed) Half-life Parent: 3 d Half-life Metabolite: 38 d Formation fraction ffMet: 0.47 page 16
Main recommendations Kinetic model • For the estimation of parameters it is necessary to identify: • Kinetic model for degradation of precursor(s), e.g. parent • SFO Vs. biphasic models • Appropriate description at least up to 10 % of the initial amount is necessary • Kinetic model for degradation of metabolite • SFO Vs. biphasic models (FOMC, DFOP) page 17
Parent FOMC Main recommendations Kinetic Model - Example 1 (Parent Degradation) DegT50 Parent: 16 d Half-life: Metabolite: 14 d Formation fraction: 0.65 Parent SFO Half-life Parent: 21 d Half-life Metabolite: 9 d Formation fraction: 1 page 18
Metabolite FOMC Half-life Parent: 1 d DegT50 Metabolite: 15 d DegT90 Metabolite: 95 d Main recommendations Kinetic Model - Example 2 (MetaboliteDegradation) Metabolite SFO Half-life Parent: 1 d Half-life Metabolite: 18 d DegT90 Metabolite: 61 d page 19
Main recommendations Weighting Method • Data weighting • Unweighted fit should be used in the 1st step • In special cases data weighting can be useful. But sufficient information for a weighting, e. g. information about the quality of data points within a data set, is usually not present • First part of the precursor’s decline curve, covering formation phase of the metabolite is more important than later time points page 20
Weighted fit (fractional) - SFO Half-life Parent: 18 d Half-life Metabolite 1: 47 d Half-life Metabolite 2: 369 d Main recommendations Weighting method - Example Unweighted fit - SFO Half-life Parent: 13 d Half-life Metabolite 1: 42 d Half-life Metabolite 2: 133 d page 21
Main recommendations Stepwise approach I • A stepwise parameter fit is recommended in the following • cases: • Complex systems with several metabolites • The pathway is not fully defined with regards to the formation of minor metabolites and bound residues • Non-SFO kinetic models are considered • Data sets with scattered or limited data points page 22
Parent Met 1 S i n k Met 2 Met 3 Main recommendations Stepwise approach II • Fit parent substance • Add primary metabolite(s), fit with parent parameters fixed to values obtained in 1), check flow to sink and simplify if justified • Fit parent and primary metabolite(s) using values obtained in 1) and 2) as starting values • Add secondary metabolite(s), fit with parent and primary metabolite(s) parameters fixed to values obtained in 3), check flow to sink and simplify if justified • ---- • Final step: fit all substances together using values obtained in n-1) as starting values page 23
Procedure to derive endpoints for metabolites Implementation of the conceptual model in a kinetic model I • Combine parent kinetics (SFO, FOMC, DFOP or other model), metabolite formation fraction and metabolite kinetics (SFO, FOMC, DFOP or other) • - Selected kinetic models must be consistent with intended use (trigger Vs. modeling) • - Use of Metabolites decision flow charts • Integrated equations with analytical solution exist for simple cases • or • Use sets of differential equations in compartment models with software tool for solving, e. g. ModelMaker page 24
Procedure to derive endpoints for metabolites Implementation of the conceptual model in a kinetic model II Parent kP* ffMet1*P kP* ffMet2*P kP*(1- ffMet1- ffMet2)*P Metabolite1 Metabolite2 kMet2* Met2 kMet1* Met1 Sink (other metabolites, bound residues, CO2) Parent: dP/dt = –kP*P Metabolite 1: dM1/dt = kP* ffMet1*P – kMet1* Met1 Metabolite 2: dM2/dt = kP* ffMet2*P – kMet2* Met2 Sink: dSink/dt = kP*P * (1 – ffMet1 – ffMet2) + kMet1* Met1 + kMet2* Met2 page 25
RUN parent only SFO, FOMC Data entry SFO fit acceptable and statistically more appropriate than FOMC no RUN parent only DFOP FOMC and/or DFOP fit acceptable? Determine best-fit model yes yes RUN parent best-fit and metabolite no see next slide Case-by-case decision Procedure to derive endpoints for metabolites Flow sheet for Trigger Endpoints PART A page 26
Procedure to derive endpoints for metabolites Flow sheet for Trigger Endpoints PART B SFO fit for metabolite acceptable? Use estimated SFO trigger endpoints (DT50 and DT90 values) yes no RUN parent best-fit and metabolite FMOC FMOC fit for metabolite acceptable? Use estimated FMOC trigger endpoints (DT50 and DT90 values) yes no Case-by-case decision page 27
RUN parent only SFO Data entry SFO fit acceptable? no Parent SFO acceptable see next slide yes RUN parent and metabolites all-SFO SFO fit for metabolites acceptable? Use estimated SFO endpoints for fate modelling yes no Case-by-case decision Procedure to derive endpoints for metabolites Flow sheet for Modelling Endpoints PART A page 28
SFO fit acceptable? no RUN parent only with appropriate biphasic model Biphasic fit acceptable? no Case-by-case decision Parent SFO non-acceptable RUN parent biphasic and metabolites all-SFO SFO fit for metabolites acceptable? Use estimated endpoints for fate modelling yes no Case-by-case decision Procedure to derive endpoints for metabolites Flow sheet for Modelling Endpoints PART B page 29
Goodness-of-fit I • Main tool for assessing goodness-of-fit: Visual assessment of • Sampling points and fitted curves • Plots of residuals • - determination that the residuals are randomly distributed • - systematic error indication that the pathway or kinetic • model used is maybe not appropriate • Overall-Fit (determination coefficient r2) • Parent and metabolites with the highest measured levels carry • more weight than metabolites at lower level • an overall fit may still appear acceptable while one or more of the metabolites may not be well fitted • For that reason, overall goodness-of-fit is not performed, • instead each substance is evaluated, separately page 30
Goodness-of-fit II • 2 test • Tool for model comparison • Tool for assessing the Goodness-of-fit of an individual substance • 2 error value should be calculated for each metabolite • (using all data used in the fit, including the sampling points below LOD or LOQ before the formation phase and after the decline phase that are included as ½ LOD or ½ (LOQ+LOD). The time-0 sample however, if set to 0 should not be used in the 2 error determination) • Error value at which the 2-test is passed for the metabolite should be below 15 % (not an absolute cut-off criterion) page 31
Goodness-of-fit III • Reliability of the individual rate parameter • Reliability of individual rate parameter estimates based on • - t-test or • - confidence intervals of the parameters • Important for metabolites that do not show a clear decline • to discern between metabolites that are persistent and • metabolites that are degrading and forming at the same • time at a similar rate page 32
Conclusions I • The half-life or DT50 value of a metabolite is not sufficient for the description of the fate of a metabolite! • Rate of formation must be considered in addition to rate of degradation • Formation and degradation are linked, and the parameters can be highly correlated • Degradation of the precursor(s) must be described properly to be able to describe the degradation of the metabolite • Number of data points for metabolites and their concentrations are often lower than for parent substances • The maximum amount and the decline phase of the metabolite are not reached during the study in some cases page 33
Conclusions II • Metabolites kinetics is more complex than for parent because formation and degradation occur simultaneously • — Complexity increases with complexity of pathway • Number of precursors (e.g. parent, metabolites) • Number of successive degradation steps • — Complexity increases with complexity of kinetic models • Formation • Degradation page 34
Conclusions III FOCUS Report: • Guidance provided for deriving metabolite kinetic endpoints from studies with parent • Trigger endpoints: degradation/dissipation DT50 and DT90 • Modeling endpoints: formation and degradation rate • Harmonized approach for reproducible results independent of software tool used • Better acceptance of generated endpoints • Facilitates review process page 35