Proton NMR Spectroscopy
690 likes | 1.11k Views
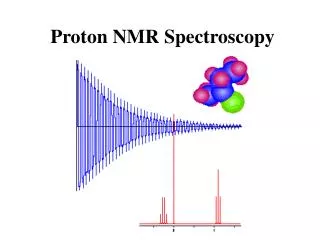
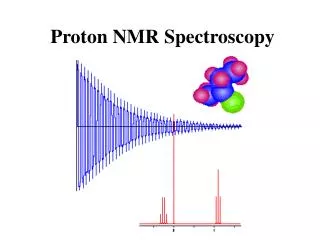
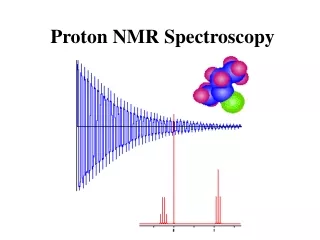
Proton NMR Spectroscopy. The NMR Phenomenon. Most nuclei possess an intrinsic angular momentum , P . Any spinning charged particle generates a magnetic field. P = [I(I+1)] 1/2 h/2 p where I = spin quantum # I = 0, 1/2, 1, 3/2, 2, …. Which nuclei have a “spin”?.

Proton NMR Spectroscopy
E N D
Presentation Transcript
The NMR Phenomenon • Most nuclei possess an intrinsic angular momentum, P. • Any spinning charged particle generates a magnetic field. P = [I(I+1)]1/2 h/2p where I = spin quantum # I= 0, 1/2, 1, 3/2, 2, …
Which nuclei have a “spin”? • If mass # and atomic # are both even, I = 0 and the nucleus has no spin. e.g. Carbon-12, Oxygen-16 • For each nucleus with a spin, the # of allowed spin states can be quantized: • For a nucleus with I, there are 2I + 1 allowed spin states. 1H, 13C, 19F, 31Pall have I = 1/2 DE = g(h/2p)Bo
When a nucleus aligned with a magnetic field, B0, absorbs radiation frequency (Rf), it can change spin orientation to a higher energy spin state. By relaxing back to the parallel (+1/2) spin state, the nucleus is said to be in resonance. Hence, NMR
NMR instruments typically have a constant Rf and a variable B0. A proton should absorb Rf of 60 MHz in a field of 14,093 Gauss (1.4093 T). Each unique probe nucleus (1H perhaps) will come into resonance at a slightly different - and a very small percentage of - the Rf. All protons come into resonance between0 and 12/1,000,000 (0 – 12 ppm) of the Rf.
Energy Difference (E) Between Two Different Spin States of a Nucleus With I=1/2
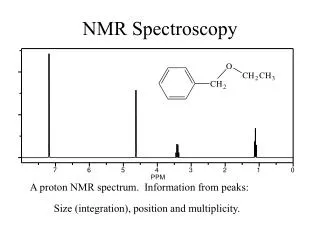
What Does an NMR Spectrum Tell You? • # of chemically unique H’s in the molecule # of signals • The types of H’s that are present e.g. aromatic, vinyl, aldehyde … chemical shift • The number of each chemically unique H integration • The H’s proximity to eachother spin-spin splitting
Chemical Shift is Affected by Electron Density Around Nucleus