Understanding Spontaneity in Voltaic Cells: E, ∆G, w, Q’, K
80 likes | 195 Views
Explore the relationship between electrode potentials, standard free energy change, work done, charge movement, and equilibrium constants in voltaic cells. Learn how to predict spontaneity in reactions and understand the dynamics of cell reactions.

Understanding Spontaneity in Voltaic Cells: E, ∆G, w, Q’, K
E N D
Presentation Transcript
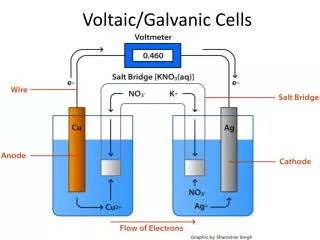
Spontaneity in Voltaic Cells Relating E, ∆G, w,Q’, K
Eored + Eoox = Eo • Higher Eored means stronger oxidizer • Higher Eoox means stronger reducer • If Eo is positive , then the reaction is spontaneous.
∆G - It’s fun • ∆G is the available free energy - or energy available for work • By convention, we identify work which is negative with work which is being done by the system on the surroundings. And negative free energy change is identified as defining a spontaneous process. ∆G = -w • Work in a voltaic cell comes from e- movement (Q’)- The movement is from charge attraction (E). • So… w = Q’E
Q’ = (9.648x104 ) (number of moles (n)) • Q’ = nF • ∆G = -w so ∆G = -Q’E and ∆G = -nF E • E is in volts, so Q’E is in (coulomb volts) which is equal to Joules (J) the unit for ∆G
Example Zn + H+ → Zn2+ + H2 Half Reactions: Zn → Zn2+ + 2e- Eoox = 0.762v H+ + 2e- → H2 Eored = 0.000v Eo = 0.762v ∆G = -nF E = -2(9.648x104 )(0.762v) ∆G = -147000J = -147kJ
K means Equilibrium Konstant • Equilibrium means the reaction will also go in reverse: A + B ↔ C + D • If the reaction goes in reverse that will change the voltage (E) and the free energy (∆G)
∆G = -RT lnK R is a constant = 8.31 T is temperature in Kelvin (K) • If ∆G = -nF E then E = • So… E = lnK
If you have standard conditions (1atm, 25oC) then we know… • So Eo lnK