Oxygen Consumption Studies in Halothiobacillus neapolitanus Using Tetrathionate and Thiosulfate
40 likes | 170 Views
This study investigates the oxygen consumption of the obligate chemolithoautotroph Halothiobacillus neapolitanus when fed with tetrathionate and thiosulfate. Using a biological oxygen monitor, we measured oxygen consumption to elucidate the metabolic pathways involved in substrate oxidation. Results indicated that 0.315 ± 0.009 µmol of oxygen was consumed with tetrathionate, while thiosulfate prompted a higher consumption of 0.192 ± 0.005 µmol. The study revealed substrate-dependent oxygen consumption patterns, contributing to the understanding of sulfur oxidation mechanisms in H. neapolitanus.

Oxygen Consumption Studies in Halothiobacillus neapolitanus Using Tetrathionate and Thiosulfate
E N D
Presentation Transcript
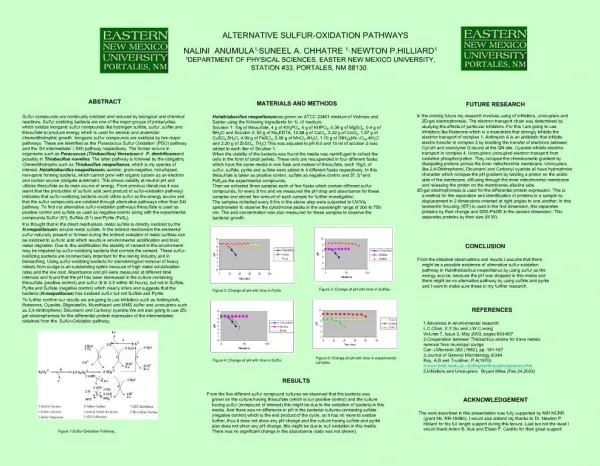
Studies on Oxygen Consumption in the Obligate Chemolithoautotroph Halothiobacillus neapolitanusRamuKakumanu*, John Latham, Neela .G, Suneel Chhatre & Newton P. Hilliard, Department of Physical Sciences, Eastern New Mexico University, Portales, NM 88130 • Oxygen consumption with tetrathionate as substrate • Utilizing tetrathionate as the substrate 0.315 ± 0.009 µmol of oxygen consumed by H. neapolitanus cells (Table 2 & Fig.3c), When tetrathionate was added to cells treated With 10 mM cyanide and azide, a reduction of oxygen consumption by ~5% was evident. Antimycin A, myxathiozol, Rotenone, and TTFA did not inhibit oxygen consumption (Fig. 3d). NEM treated cells were completely inhibited and yielded a 0% change in O2 content. • Methods & Materials • Cell culture and colony purification was done by transferring H. neapolitanus DSMZ 15147 to culture flask containing a two solution media as described by Canter/Vishniac and modified by Jones. • Large scale growth of H. neapolitanus was accomplished in a 10L chemostat. The effluent from the fermentor was collected in a carboy held at 00C. The cells were harvested from the medium by centrifugation at 15,000g and stored at 00C until used. Background • Halothiobacillus neapolitanus is an aerobic, gram- negative, obligate chemolithoautotroph, which oxidizes reduced sulfur compounds such as sulfide, thiosulfate, sulfur, tetrathionate for energy, furthermore it has optimal growth at 30-400C and can proliferate at pH 4.5. • To utilize H.neapolitanus, in ongoing enzymatic research and proteins thereof for commercial use it is necessary to elucidate its metabolic pathway, substrate to oxygen ratios and end products of substrate oxidation. • Inorganic sulfur compounds are oxidized by two major pathways. These are identified as the PSO pathway that occurs in organisms such as Paracoccus versutus and P. dentrificans and the S4I pathway which has been proposed as the oxidation route for H. neapolitanus. • Oxygen consumption measurement • Oxygen consumption was measured at 300C using an YSI 5300A Biological Oxygen Monitor and well connected a computer via serial port. • To the wells 2ml of 30Mm MOPS and 120Mm KCL PH 6.8 and 0.96ml of deionized H2O was added, 30µl of 0.047 mg/ml wet weight whole cells suspended in the MOPS buffer solution was added to the well. • Once % O2 stabilized, 10 µl of stock solution of substrate consisting of either thiosulfate or tetrathionate in water was added via Hamilton syringe and air consumption was determined. • Cell solution was filtered via 0.2 µm membrane filter, protein concentration was measured via Bio Rad Protein assay kit. Lane 1: Molecular Marker Lane 2: End-filled DNA before ligation Sulfur oxidation pathways • Fig.1. shows the S4I pathway in H.neapolitanus which includes multi-sulfur compounds such as thiosulfate, tetrathionate and trithionate as intermediates. • However, this pathway remains only as a proposal due to inconclusive evidence. O2 consumption, studies will allow us to validate alternative S4I pathway. • Fig.2. shows PSO pathway which does not include multi-sulfur compounds as intermediates. Since this pathway does not include tetrathionate as intermediate it can be eliminated as the primary pathway. Fig.1. Proposed Sulfur Oxidation pathway in H. neapolitanus Fig.3. Oxygen consumption by H. neapolitanus using (Clockwise from top left) a) thiosulfate, b) thiosulfate and inhibitors, c) tetrathionate and inhibitors, and d) tetrathionate • Conclusion • Oxygen consumption was found to be substrate dependent for the substrates investigated, maximum O2 consumption occurred when utilizing thiosulfate as substrate, the substrate and oxygen ratios were found to be 1:2 and 1:3. • Substrate to oxygen ratios imply that thiosulfate oxidation is stoichiometrically balanced with oxygen consumption where as tetrathionate oxidation may not be, it is also evident that H.neapolitanus does not utilize the same glucose based electron transport system. • Results • Oxygen consumption with thiosulfate as the substrate • H. neapolitanus cells consumed 0.192 ± 0.005 µmol of oxygen (Table1 & Fig.3a) when thiosulfate was injected into the reaction well. • The cells were sensitive to 10 mM cyanide treatment with a reduction of oxygen consumption (Fig.3b),azide treatment affected the rate of oxygen consumption of H. neapolitanus. • The treatment of cells with 0.8 mM and 0.04 mM NEM resulted in a reduction of total oxygen consumption.Conversely, antimycin A, myxathiozol, rotenone, and TTFA did not display any inhibition via rate or total consumption towards the metabolic oxidation of thiosulfate. Fig.2. Proposed Thiosulfate oxidation pathway in P.pantotrophous • The aim of this study is to measure the oxygen consumed by H. neapolitanus cells during substrate oxidation through S4I pathway using tetrathionate, thiosulfate as substrates. • The consumed oxygen was measured using an oxygraph apparatus which utilizes a clark type polarographic oxygen electrode. • From this study we investigated the oxygen consumed via oxygraph of thiosulfate and Tetrathionate to establish a stiochiometric ratio of substrate to molecular oxygen. In addition inhibitors such as cyanide, azide, TTFA, ampicillin A, and myxathiozol were used to determine if H. neapolitanus utilizes the typical four complex ETS. • Future Research • The future experiments with oxygraph and ion-chromatography techniques will enable us to detect changes in metabolism in H. neapolitanus as the pH of the growth media is lowered. Acknowledgements The work described in this presentation supported by NIH NCRR grant number R-16480. We thank John Latham, Joaquin Deleon for their great support and experimental assistance. • Kelly, D.P., Shergill, J.K., Lu, W.P., Wood, A. P. Oxidative metabolism of inorganic sulfur compounds by bacteria. Antonie van Leeuwenhoek. 71 (1997) 95-107. • Trudinger, P. A. The effects of Thiosulphate and Oxygen Concentration on Tetrathionate Oxidation by Thiobacillus X and T. thioparus. Biochemical Journal. 90 (1964) 640-646. • Masau, R. J. Y., Oh, J. K., and Suzuki, I. Mechanism of oxidation of inorganic sulfur compounds by thiosulfate-grown Thiobacillus thiooxidans. Canadian Journal of Microbiology. 47 (2001) 348-358. • YSI 5300A Biological Oxygen Monitor. Accessed dec 2008. http://www.ysilifesciences.com/extranet/BTKL.nsf/447554deba0f52f2852569f500696b21/dbf5af3c473da1dc85256ba4007bb5ec!OpenDocument • YSI Biological Oxygen Monitor System Operations Manual. A530035 Rev A. February 2002. • References • Friedrich, G. C. Physiology and Genetic of Sulfur Oxidizing Bacteria. Advanced Microbial Physiology. 39 (1998) 235-238. • Visser, J. M., Robertson L. A., Van Verseveld H. W., Kuenen, J. G. Sulfur Production by Obligately Chemolithoautotrophic Thiobacillus Species. Applied and Environmental Microbiology. (1997) 2300-2305. • Kelly, D.P., Wood, A. Reclassification of some species of Thiobacillus to the newly designated genera Acidothiobacillus gen. nov., Halothiobacillus gen. nov. and Thermithiobacillus gen. nov. International Journal of Systematic and Evolutionary Mircrobiology. 50 (2000) 511-516. • Trudinger, P. A. Oxidation of thiosulphate by intact cells of Thiobacillus X: Effects of some experimental conditions. Australian Journal of Biological Science 17 (1964) 738–751. • Lu W.P., Swoboda, B.P. and Kelly, D.P. Properties of the thiosulphate-oxidizing multi-enzyme system from Thiobacillus versutus. Biochimica et Biophysica Acta. 828(1985) 116–122.