Acids & Bases
370 likes | 396 Views
Learn about acids and bases, their properties, and their presence in everyday life. Discover how they interact, their effects on substances, and the significance of pH levels. Presented by Dr. Seemal Jelani.

Acids & Bases
E N D
Presentation Transcript
Acids&Bases They are everywhere.. In your food In your house EVEN IN YOU!!!!! Dr Seemal Jelani
What is an acid? • An acid is a solution that has an excess of H+ ions. It comes from the Latin word acidus that means "sharp" or "sour". • The more H + ions, the more acidic the solution. Dr Seemal Jelani
Properties of an Acid • Tastes Sour • Conduct Electricity • Corrosive, which means they break down certain substances • Many acids can corrode fabric, skin, and paper Dr Seemal Jelani
Introduction to Acids (con’t) • Lactic acid is also produced by bacteria in milk – this is why milk turns sour. • Acids occur naturally in many fruits. • Insects such as millipedes, scorpions, and ants use acids to deter predators. • A physician may use a solution of boric acid to rinse out your eyes, but a drop or two of many other acids would blind you. • Some acids add a tangy, sour flavour to foods and drinks, while others are deadly.
Some acids react strongly with metals • Turns blue litmus paper red Dr Seemal Jelani
Definitions Arrehenius only in water • Acids – produce H+ • Bases - produce OH- • Acids – donate H+ • Bases – accept H+ • Acids – accept e- pair • Bases – donate e- pair Bronsted-Lowry any solvent Lewis used in organic chemistry, wider range of substances Dr Seemal Jelani
The hydrogen ion in aqueous solution Examples H+ + H2O H3O+ (hydronium ion) Arrhenius HCl NaOH Bronsted-Lowry HCl HCN NH3 :NH3 Lewis BF3 Dr Seemal Jelani
Word Connect • The word “acid” comes from the Latin acidus, meaning “sour” • Think of the taste of fruits such as lemons or limes • These fruits contain citric acid • Vinegar, a common household product, contains acetic acid.
Chemists sometimes use the term “alkali” for a base that is soluble in water • This word shows the Arabic origin of chemistry • The earliest source of bases was the ash obtained by burning plants • Thus the Arabic word alkali means “ashes of a plant.” Dr Seemal Jelani
Alkali salts are soluble hydroxides of alkali metals and alkaline earth metals, of which common examples are: • Sodium hydroxide – often called "caustic soda" • Potassium hydroxide – commonly called "caustic potash" • Lye – generic term for either of the previous two or even for a mixture • Salts of alkali • Reaction with water • What is an example of a strong alkali?(pH 14) Dr Seemal Jelani
Strong acids and weak acids • High concentration of Hydrogen ions • Low concentration of Hydrogen ions • What is difference between base and alkali? • Bases are substances that react with acids and neutralize them. • They are usually metal oxides, metal hydroxides, metal carbonates or metal hydrogen carbonates. Many bases are insoluble - they do not dissolve in water. If a base does dissolve in water, we call it an alkali. Dr Seemal Jelani
What is an alkaline? • The “alkaline” in alkaline water refers to its pH level. The pH level is a number that measures how acidic or alkaline a substance is on a scale of 0 to 14 • Examples: acids, base, alkaline(pH > 7.0) Dr Seemal Jelani
An alkaline solution is a mixture of base solids dissolved in water. • The potential of hydrogen, also known as the pH scale, measures the alkalinity or acidity level of a solution. • The scale ranges from zero to 14. The midpoint 7 represents a neutral pH. • A neutral solution is neither an acid nor alkaline. pH levels below 7 indicate an acidic solution, and numbers above 7 indicate an alkaline solution. • The pH is a measurement of intensity, not capacity Dr Seemal Jelani
Is alkaline a base or acid? • The solution is neither acidic or basic. ... Because the base "soaks up" hydrogen ions, the result is a solution with more hydroxide ions than hydrogen ions. • This kind of solution is alkaline. • Acidity and alkalinity are measured with a logarithmic scale called pH. Dr Seemal Jelani
pH • pH is the measure of acidity or alkalinity of a solution on a logarithmic scale on which 7 is neutral, lower values are more acid and higher values more alkaline. • The pH is equal to −log10c, where c is the hydrogen ion concentration in moles per litre. • The pH in the chemistry test used to test the acidity of substances stands for 'potential hydrogen,' Dr Seemal Jelani
What are pH levels? • why is it important? • The importance of pH. The pH level of water measures how acidic it is (pH stands for potential hydrogen, referring to how much hydrogen is mixed with the water.) 7 is a balanced pH for water. Anything below 7 indicates the water is acidic, and if it's above 7 it is alkaline. Dr Seemal Jelani
What should your body pH be? • Those levels vary throughout your body. • Your blood is slightly alkaline, with a pH between 7.35 and 7.45. • Your stomach is very acidic, with a pH of 3.5 or below, so it can break down food. Dr Seemal Jelani
Why is pH so important to life? • Acids and bases are important in living things because most enzymes can do their job only at a certain level of acidity. • Cells secrete acids and bases to maintain the proper pH for enzymes to work. ... • The acidic environment helps with the digestion of food Dr Seemal Jelani
What does a pH of 5.5 in urine mean? • A neutral pH is 7.0. The higher the number, the more basic (alkaline) it is. • The lower the number, the more acidic your urine is. • The average urine sample tests at about 6.0. If your urine sample is lower, this could indicate an environment conducive to kidney stones. Dr Seemal Jelani
Uses of Acids • Acetic Acid = Vinegar • Citric Acid = lemons, limes, & oranges. It is in many sour candies such as lemonhead & sour patch. Dr Seemal Jelani
Ascorbic acid = Vitamin C which your body needs to function. • Sulfuric acid is used in the production of fertilizers, steel, paints, and plastics. • Car batteries Dr Seemal Jelani
What is a base? • A base is a solution that has an excess of OH- ions. • Another word for base is alkali. • Bases are substances that can accept hydrogen ions Dr Seemal Jelani
Bases are bitter-tasting compounds with a slippery feel. Most soaps and drainers and window cleaners are bases.
Properties of a Base • Feel Slippery • Taste Bitter • Corrosive • Can conduct electricity. (Think alkaline batteries.) • Do not react with metals. • Turns red litmus paper blue. Dr Seemal Jelani
Uses of Bases • Bases give soaps, ammonia, and many other cleaning products some of their useful properties. Dr Seemal Jelani
The OH- ions interact strongly with certain substances, such as dirt and grease. • Chalk and oven cleaner are examples of familiar products that contain bases. • Your blood is a basic solution. Dr Seemal Jelani
Defining Indicators • Two of the most common indicators are phenolphthalein and litmus. • Litmus is a compound that is extracted from lichens, a plant-like member of the fungi kingdom • Litmus paper is made by dipping paper in litmus solution.
Chemical indicator, any substance that gives a visible sign, usually by a colour change. • What are indicators give examples? • Acid-Base Indicator Examples. • The best known pH indicator is litmus. Thymol Blue, Phenol Red and Methyl Orange are all common acid-base indicators. • Red cabbage can also be used as an acid-base indicator
What are natural indicators give examples? • An acid (e.g. vinegar, lemon juice), purple in an alkali (e.g. bicarbonate of soda, bleach) and green in something that is neutral (e.g. water). • Many plants contain their own indicators – turmeric, red cabbage juice and beetroot juice are three good examples. Other examples are tea and red grape juice. Dr Seemal Jelani
How do indicators work in chemistry? • pH indicators detect the presence of H+ and OH-. • They do this by reacting with H+ and OH-: they are themselves weak acids and bases. • If an indicator is a weak acid and is coloured and its conjugate base has a different colour, deprotonation causes a colour change. Dr Seemal Jelani
What are some examples of indicators? Some Common Indicators Dr Seemal Jelani
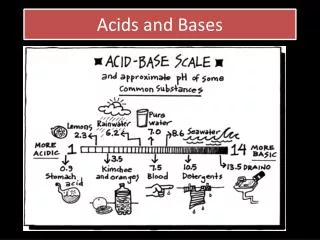
pH Scale • pHis a measure of how acidic or basic a solution is. • The pH scale ranges from 0 to 14. • Acidic solutions have pH values below 7 • A solution with a pH of 0 is very acidic. • A solution with a pH of 7 is neutral. • Pure water has a pH of 7. • Basic solutions have pH values above 7. Dr Seemal Jelani
Acid – Base Reactions • A reaction between an acid and a base is called neutralization. An acid-base mixture is not as acidic or basic as the individual starting solutions. Dr Seemal Jelani
Acid – Base reactions • Each salt listed in this table can be formed by the reaction between an acid and a base. Dr Seemal Jelani
Neutralization In general: Acid + Base Salt + Water All neutralization reactions are double displacement reactions. HCl + NaOH NaCl + HOH HCl + Mg(OH)2 H2SO4 + NaHCO3 Dr Seemal Jelani
Strong and Weak Acids/Bases Strong acids/bases – 100% dissociation into ions HCl NaOH HNO3 KOH H2SO4 Weak acids/bases – partial dissociation, both ions and molecules CH3COOH NH3 Dr Seemal Jelani
- H+ + H+ When life goes either way amphoteric (amphiprotic) substances Acting like a base Acting like an acid HCO3- H2CO3 CO3-2 accepts H+ donates H+ Dr Seemal Jelani