Chapter 4 Chemical Reactions
Chapter 4 Chemical Reactions. HW #3 Due by Monday, 5/5/2014 HW #4 due by Friday, 5/9/2014 Quiz #2: Monday, 5/12/2014 Exam #2: Wednesday, 5/14/2014. End-of-Chapter Homework: pp 167 - 177. 5, 6, 9-11 14-18 19 (a,d,g,h) 28 29 31 32 33 38 41 43

Chapter 4 Chemical Reactions
E N D
Presentation Transcript
Chapter 4Chemical Reactions HW #3 Due by Monday, 5/5/2014 HW #4 due by Friday, 5/9/2014 Quiz #2: Monday, 5/12/2014 Exam #2: Wednesday, 5/14/2014
End-of-Chapter Homework: pp 167 - 177 5, 6, 9-11 14-18 19(a,d,g,h) 28 29 31 32 33 38 41 43 47 51 55 57 59 61 65b 67 69 71 73 79 81 83 89 100
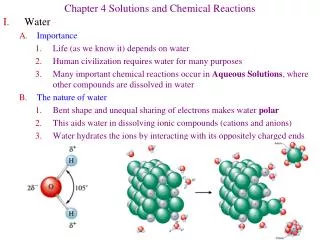
I. Solubility - Many Ionic compounds dissolve in water yielding ions. -These soluble ionic compounds conduct electricity due to the ions. Are called electrolytes. - Driving force is water being attracted to the ions, forming weak bonds, & releasing energy. 1) Nonelectrolyte (does not conduct electricity in water): a) Molecular & Soluble - when dissolves, yields no ions - CH3OH b) Ionic & Insoluble like AgCl or PbI2 2) Strong Electrolyte (Ionic, Soluble, Conducts well in water) NH4Cl(s) -----) NH4+(aq) + Cl-(aq) 3) Weak Electrolyte (few % ionization in water; Weak Conductor) NH3 + H2O ------) NH4+ + OH- (~ 1% ionized) HC2H3O2 --------) H+ + C2H3O2- (~ 1 % ionized)
Svante Arrhenius NaI in H2O – Soluble & Ionic; Many Ions H2O – Molecular & No Ions Nonelectrolyte – No Current Electrolyte – Much Current
I. Solubility Continued • Know general solubility rules : These allow you to predict both the solubility & products of many Reactions (Rxns). #1 Grp 1, NH4+, C2H3O2- &NO3- compds are soluble (except H). #2 Cl- Br- I-compds are solubleexcept : Ag+ Pb2+ Hg22+ #3 SO4-2solubleexcept : Ca2+ Sr2+ Ba2+ Ag+ Pb2+ Hg22+ #4 OH-compds are insolubleexcept#1 & Ca2+ Sr 2+ Ba 2+ #5 Most CO3-2 (carbonates), PO4-3 (phosphates), & S-2 (sulfides) are insoluble except for #1, (rule 1).
II. Writing Reactions - Ways to write; Example 1 [Note: Ag ion is always Ag+1 does not need (I); Solvent is H2O] silver nitrate + sodium chloride --------) silver chloride + sodium nitrate 1) Molecular Equation (ME):Write all as if not ionized AgNO3(aq) + NaCl(aq) --------) AgCl(s) + NaNO3(aq) 2) Complete Ionic Equation (CIE):Write soluble, ionic compounds as ions; should use subscripts like (aq) Ag+ + NO3- + Na+ + Cl- ----) AgCl(s) + Na+ + NO3- 3) Net Ionic Equation (NIE):Eliminate Spectator Ions Ag+(aq) + Cl-(aq) -----) AgCl(s)
III. Reaction Types A. Precipitation Reactions - Formation of an insoluble compound (precipitate, ppt) is a driving force in a chemical reaction. - Can predict a product from the solubility rules. Method:Write soluble ionic reactants as ions. Check solubility rules & see if insoluble product forms from ions. Example: Predict product when mix K2SO4(aq) & BaCl2(aq) Write CIE & NIE. 2K+ + SO42- + Ba2+ + 2Cl- ---) BaSO4(s) + 2K+ + 2Cl-(CIE) Ba2+(aq) + SO42-(aq) -----) BaSO4(s)(NIE)
III. Reaction Types B. Acid-Base Rxns 1. Definitions (Note: Proton = H+) Arrhenius:Acid =substance which releases H+ in water. Base =substance which releases OH- in water. Examples: HCl(aq) -----) H+(aq) + Cl-(aq) NaOH(aq) -----) Na+(aq) + OH-(aq) Bronsted-Lowry:Acid = Proton Donor Base = Proton Acceptor Example:(air is the solvent) NH3(g) + HCl(g) -----) NH4Cl(s)
III. Reaction Types B. Acid-Base Rxns 2. Properties Note: salt = any ionic compound Acids: - Sour taste - Turn Indicator dyes a specific color: Phenolphthalein colorless; Litmus red - React with bases to yield salt + H2O Bases: - Bitter Taste, Feel Slippery - Turn Indicator dyes a specific color: Phenolphthalein red; Litmus blue - React with acids to yield salt + H2O - Strong Acids/Bases ionize 100% in water - Weak Acids/Bases ionize < < 100% in water
III. Reaction Types B. Acid-Base Rxns 3. Examples (Know these strong & weak acids & bases) Strong Acids:HCl HBr HI HNO3 H2SO4 HClO4 Strong Bases: NaOH KOH Ba(OH)2 (but not too sol.) Example in water: HCl(aq) -----) H+ + Cl-100% to the right Weak Acids: HF HC2H3O2 HCN H2CO3 Weak Bases: NH3[= NH4OH (in water)]NaHCO3 Examples in water: NH4OH NH4+ + OH-~1% to the right HC2H3O2H+ + C2H3O2-~1% to the right
III. Reaction Types B. Acid-Base Rxns 4. Acid-Base or Neutralization Reactions - Acids react with bases to give: salt(ionic compd) + H2O - Driving force is to produce stable H2O& sometimes a gas. - Examples:NaOH + HF -----) NaF + H2O 2KOH + H2SO4 ---) K2SO4 + 2H2O • Some Neutralization Rxns produce a gas:(memorize these six) H+ + HCO3- -----) CO2(g) + H2O 2H+ + CO32------)CO2(g)+ H2O 2H+ + SO32- -----) SO2(g) + H2O 2H+ + S2- -----) H2S(g) H+ + CN- -----) HCN(g) NH4+ + OH------) NH3(g)+ H2O
III. Reaction Types B. Acid-Base Rxns 5. Write three rxn formats (ME, CIE & NIE) for reaction of potassium hydroxide with hydrochloric acid: ME:KOH + HCl -----) KCl + H2O CIE:K+ + OH- + H+ + Cl- -----) K+ + Cl- + H2O NIE:H+(aq) + OH-(aq) -----) H2O(l) Note: 1) The above net ionic equation is common for many acid-base reactions. The production of stable H2O is a driving force for the reaction. 2) H+ in water is better represented by H3O+ 3) H3O+is a polyatomic ion = hydronium ion
III. Reaction Types B. Acid-Base Rxns 5. ME, CEI and NIE - Write the ME, CIE & NIE for: HCl + NaHCO3(in water) ME: HCl + NaHCO3 -----) NaCl + H2O + CO2 CIE: H+ + Cl- + Na+ + HCO3- ---) Na+ + Cl- + H2O + CO2 NIE: H+(aq) + HCO3-(aq) -----) H2O(l) + CO2(g) 6. Titration - Process of adding known (amount & concentration) of Base (or Acid) in a burette to an unknown concentration of Acid (or Base). End point is determined with an indicator or a pH meter. Can now determine the amount of the unknown (quantitative analysis).
III. Reaction Types B. Acid-Base Rxns Known M & Volume (Standard Solution) + indicator After titn can calculate moles & M.
III. Reaction Types C. Predicting products - Review • Predicting products:write a) strong acids & b) solubleionic reactants as ions; look to see if the ions can form: (1) aprecipitate(solubility rules) (2) agas(six gas equations) (3) anonionized molecule like H2O or HC2H3O2 • Write NIE (in water) for: 1) BaI2 + AgNO3 & 2) HCl + NaCN 1) Ba+2 + I- + Ag+ + NO3- -----) AgI(s) + Ba+2 + NO3- Ag+(aq)+ I-(aq) -----) AgI(s)(Balanced NIE) 2) H+ + Cl- + Na+ + CN- -----) HCN(g) + Na+ + Cl- H+(aq) + CN-(aq) -----) HCN(g)(Balanced NIE)
III. Reaction Types D. Redox Rxns 1. Definitions & Introduction: - Oxidation Reduction Reaction (Redox Rxn) is one in which electrons are transferred. - Oxidation = Loss of Electrons - Reduction = Gain of Electrons 2. Examples: (Note: electrons lost & gained must be same) 2Mg + O2 -----) 2MgO [ 2Mgo ---) 2Mg2+ + 4e- = Oxidation ] Half Rxn [ O2o + 4e- -----) 2O2- = Reduction ] Half Rxn 3Cu+2 + 2Al -----) 3Cu + 2Al+3 [ 2Alo -----) 2Al+3 + 6e- = Oxidation] Half Rxn [ 3Cu+2 + 6e- -----) 3Cuo = Reduction] Half Rxn
III. Reaction Types D. Redox Rxns Note: Oxidizing agent causes oxidation Reducing agent causes reduction 3. Uses of RedoxRxns: a) Create new chemicals b) Theoretical Importance (chem 123) c) Battery – allow the e’stransferred to perform work 4. Oxidation Numbers (ON): Definition:ON = the charge that an atom or group of atoms would have if they were ionic. Uses:1)Redox Rxn(ID & what’s been oxid/red & #e-) 2) NomenclatureFeF2 = Iron(II)Fluoride; (II) = ON 3) Used in balancing redox rxns(chem 123) Rules: Know the following rules [Order gives priority]
III. Reaction Types D. Redox Rxns 5. Rules for Determining ON (Know) Note: When is a conflict, 1st rule controls result. a) The sum of ON’s add up to give the charge b) The ON of a neutral element by itself is 0 c) Group 1, 2, 13 ions are +1, +2 & +3 respectively; (exception: H is -1 when combined with metal: KH , NaH) d) F is -1 e) O is -2 (exception: O is -1 when found as a peroxide, O22-) f) Cl, Br, I are -1 [when together like BrCl, most electronegative = -1]
III. Reaction Types D. Redox Rxns Examples:Determine the ON of single N in each: N2 N3-1 Na3N N2O NO NF3 NO2 NO3-1 0 -1/3 -3 +1 +2 +3 +4 +5 - Which of the above forms of N can explosively react with organics? Why? - Determine ON of each atom in FeSO3(Note: SO3-2 ON = -2) [+2 +4 -2] • Which are redox: (ones with change in ON; ∆ON) 1) Mg + Cl2 -----) MgCl2 2) CH4 + 2 O2 -----) CO2 + 2 H2O 3) HCl + NaOH -----) H2O + NaCl
III. Reaction Types D. Redox Rxns 6. Redox Reaction Types Combination: 2Al + 3F2 -----) 2AlF3 Decomposition: 2HgO -----) 2Hg + O2 Combustion: CH4 + 2O2 -----) CO2 + 2H2O Single Replacement: 2Na + 2HCl -----) 2NaCl + H2 [ Can predict reaction from table 4.6, Pg 153; The more active metal will replace the cation ] 7. Balancing redox rxns: Can become difficult; so, rules were generated & will cover in Chemistry 123.
IV. Quantitative Aspects of Solutions A. Molarity, M - Many reactions take place in solution & we need a way to measure concentration; Mis common way. - M = moles of Solute M = m (use as eqn) L Solution L solvent = substance present in largest amount solute = substances dissolved in solvent solution = solute + solvent (everything present) B. Molarity Calculations 1) Calculate the M if 4.5 moles of HCl are dissolved in a total volume of 500 mL (0.500 L) M = m = 4.5 moles = 9.0 mHCl = 9.0 MHCl L 0.500 L L
IV. Quantitative Aspects of Solutions 2) Calculate M if 0.020 g of CaCO3 are dissolved in 40 L a) convert g CaCO3to mols; b) plug into M eqn; M = m/L 0.020 g CaCO3 x 1 mole CaCO3/100. g CaCO3 = 2.0x10-4 mol CaCO3 M = moles = 2.0x10-4 mol CaCO3 = 5.0x10-6M CaCO3 L 40 L 3) How many moles of HCl are present in 2.0 L of 0.30 MHCl? M = moles/Lmoles = M x L moles= 0.30 m HCl x 2.0 L = 0.60 moles HCl L 4) How many L of 12 MHCl will provide 2.0 moles of HCl ? M = m L = m = 2.0 mole HCl = 0.17 L HCl LM 12 mole/L
IV. Quant. Aspects of Solutions C. Dilution Problems - Frequently we are given a concentrated solution & we need to add solvent to get a more dilute solution. This is a dilution problem. M1 x V1 = M2 x V2 Note: 1) Initial moles = Final moles 2) Works with any conc. or vol. units 3) Use eqn ONLY for dilution problem Example: How many mL of 12 M HCl are needed to make 100. mL of 3.0 M? M1 x V1 = M2 x V2 V1 = M2 x V2= (3.0 M x 100. mL) = 25 mL M112 M - Take 25 mL of 12 M HCl and add enough water to make 100 mL.
IV. Quantitative Aspects of Solutions D. Gravimetric Analysis - A type of analysis where one converts the analyte to an insoluble compound which is then isolated, dried & weighed. Example: A 1960 dime (2.50g) is dissolved in nitric acid. HCl is then added to form insoluble AgCl, which is collected, washed, dried & weighed. Calculate a) the g of Ag & b) w/w % Ag in the dime if 2.98 g of AgCl was obtained. Ag ---) Ag+1 ---) AgCl a) g Ag in dime: 2.98 g AgCl 1 mol AgCl 1 mol Ag 108 g Ag = 2.25 g Ag 143 g AgCl1 mol AgCl1 mol Ag b) % Ag in dime: w/w % Ag = g Ag x 100% = 2.25 g Ag x 100% = 90.0% g Dime 2.50 g
IV. Quantitative Aspects of Solutions E. Volumetric Analysis - An analysis technique where the M & volumeof a reagent in a burette is used to calculate the mols or M of an unknown. - NOTE: This is a typical stoichiometric problem except that we calculate mols of reagent from M & Linstead of from grams. Example: 25.0 mL of HCl is titrated with 45.5 mL of 0.433 MNaOH. Calculate the a) mols of HCl present & b)M of the original HCl. 1 HCl + 1 NaOH -----) 1 NaCl + 1 H2O a) m 0.433 m NaOH 0.0455 L NaOH 1 m HCl = 0.0197 m HCl L NaOH 1 m NaOH b) M = moles HCl = 0.0197 m HCl = 0.788 M HCl L 0.0250 L
E. Vol Anal, Example: Note HC2H3O2 = HA, MW = 60.05 g/m 1) Moles HA (12.1 mL NaOH = 0.0121 L) 0.500 m NaOH x 0.0121 L = 0.00605 m NaOH L 0.00605m NaOH x 1m HA/1m NaOH = 0.00605 mHA 2) M HA (10.0 mL HA = 0.0100 L HA) M= m = 0.00605 m HA = 0.605 m/L HA L 0.0100 L HA 3) w/w % HA = (g HA / g Soln) x 100% g HA = 0.00605m x 60.05 g/m HA = 0.3633 g HA w/w% HA = (0.3633g HA /10.3 g soln) x100 = 3.53%
General Example • Give the NIE for reaction of Lead (II) Nitrate with hydroiodic acid. Pb(NO3)2 + HI ----) Pb+2 + NO3- + H+ + I- ----) PbI2(s) + H+ + NO3- Pb+2(aq) + 2I-(aq) ----) PbI2(s) -? Mols PbI2 from 0.10 m HI. 0.10 m I-1 m PbI2 = 0.050 m PbI2 2 m I- - ? Mols PbI2 from 2.0 L of 0.050 m/L HI? 0.050 m HI 2.0L 1 m PbI2 = 0.050 m PbI2 L 2 m HI
Chapter 3 & 4 Review • Mole • % Composition • EF & MF • Stiochiometry: Reg, LR, Amt. Left, % Yield • Predicting Products: Solubility, Gas, H2O & Weak Acids • Writing Rxns: ME CIE NIE Phases • Electrolytes • Acids & Bases • Redox Oxidation & Reduction • ON • MM & Stiochiometry & Titrations; Dilution