Sigma ( σ ) Bonding
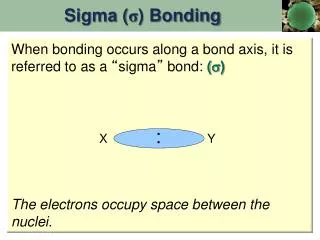
:. Sigma ( σ ) Bonding. When bonding occurs along a bond axis, it is referred to as a “ sigma ” bond: ( ) The electrons occupy space between the nuclei. X. Y. :. X Y. Pi ( π ) Bonding.

Sigma ( σ ) Bonding
E N D
Presentation Transcript
: Sigma (σ) Bonding When bonding occurs along a bond axis, it is referred to as a “sigma” bond: () The electrons occupy space between the nuclei. X Y
: X Y Pi (π) Bonding When bonding occurs above and below a bond axis, it is referred to as a “pi” bond: () The electrons occupy space above and below the nuclei.
: X Y : Sigma (σ) Bonding & Pi (π) Bonding Sigma () bonding: When bonding occurs along a bond axis, it is referred to as a “sigma” bond. Pi () bonding: When bonding occurs above and below a bond axis, it is referred to as a “pi” bond. A double bond is made up of a and a bond.
Hybridization of Atomic Orbitals The simple model of atomic orbital overlap in H2, HF and F2 breaks down for more complicated molecules. Consider methane: VSEPR theory predicts bond angles of 109.5°. These angles can’t be achieved with the s, px, py & pz orbitals of the central carbon atoms.
2p 2s carbon Hybridization of Atomic Orbitals In order to attain the needed geometry, the atomic orbitals (AO’s) mix orhybridizeto form new valence bondorbitals. consider carbon as a central atom in a molecule: The valence orbitals are the 2s & 2p’s There are 4 valence electrons:
Hybridization of Atomic Orbitals This hybridization determines by the electron pair geometry for the central atom. Each half-filled orbital is capable of forming a covalent bond. The new orbitals are called sp3 Valence bondorbitals 2p carbon 2s carbon The 4 valence electrons on carbon fill the orbitals by Hund’s rule:
sp3Hybridization Conclusion: When the central atom in a molecule has combination of 4 total sigma (single) bonds and lone pairs, the hybridization at the central atom is sp3.
sp2Hybridization Comes from mixing one ____________ AO with two _____________ Aos. They hybridize to form new valence bondorbitals or “molecular orbitals (MOs) there is one p-orbital left over 2p The new hybrid obitals all have the same energy: sp2 The new orbitals are called sp2Valence bondorbitals
sp2Hybridization The remaining p-orbital is perpendicular to the three sp2 valence bond orbitals that spread out in a plane.
sp2Hybridization Bonding in BF3 The atomic orbitals on the central B-atom can’t accommodate 3 bonds!
Bonding in BF3 The 1 s orbital and 2 p orbitals must mix to form 3 new sp2 hybrid orbitals. The 3 sp2 hybrid orbitals can now form sigma bonds with each half-filled p-orbital on each fluorine atom. This results in a “Trigonal Planar” molecular and electron pair geometry.
sp2Hybridization Bonding in an sp2hybridized atom is shown below: Each of the three sp2 orbitals can form a -bondwith another atom. Two of the orbitals overlap along the bond axis: “end on” overlap The left over p-orbital can form a -bondwith another half-filled p-orbital. results in a -bond! “Sideways” overlap...
sp2Hybridization Bonding in an sp2hybridized atom is shown below: Each of the three sp2 orbitals can form a -bondwith another atom. Two of the orbitals overlap along the bond axis: “end on” overlap The left over p-orbital can form a -bond with another half-filled p-orbital. results in a -bond! “Sideways” overlap...
sp2Hybridization An example of sp2 hybridization is given by C2H4 (ethene) The left over p-orbitals on each carbon overlap to form the -bond (second half of the double bond). Each sp2 orbital can form a -bond, two with each of the H’s and one with the other carbon. 2p 2p Carbon sp2 hybrid valence bond orbitals. H H Trigonal planar EPG at each carbon: sp2hybridizaton! C C 2s H H
-Bonding in C2H4 The unused p orbital on each C atom contains an electron and this p orbital overlaps the p orbital on the neighboring atom to form the π bond.
Consequences of Multiple Bonding There is restricted rotation around C=C bond.
Consequences of Multiple Bonding Restricted rotation around C=C bond.
Other Examples of Molecules with sp2: CH2O Conclusion: When a central atom has a trigonal planarelectronic geometry (EPG), it is most likely to bond through sp2 hybridization. Compounds containing double bonds ( + ) most often have have sp2 hybridization.
2p 2p sp hybrid valence bond orbitals form. 2s sp Hybridization A mix of ________________ and ___________ . The sp orbitals spread out to form a linear geometry (directed away from one another) leaving the unused p-orbitals perpendicular to the molecular axis. The sp orbitals can form -bonds or hold lone pairs. The two unused p-orbitals can form the (2) -bondsin a triple bond. An example of sp hybridization is given by C2H2 (acetylene) Carbon
sp Hybridization Just as with sp2 hybridization, in sp hybridization, the left over p-orbitals can form -bonds (in this case 2). In acetylene (C2H2) there is a triple bond. (1 , 2 ’s)
sp Hybridization Other examples of molecules with sp hybridization are: N2 :NN: CN– (cyanide anion) [:CN:]– Conclusion: When a central atom has a linear electronic geometry (EPG) with no lone pairs , it is most likely to bond through sp hybridization. Compounds containing triple bonds ( + 2) or adjacent double bonds (CO2) have sp hybridization.
sp, sp2, & sp3 hybridization ….has 2 leftover orbitalsavailable for triple bonding. ….has 1 leftover orbitals available for double bonding. ….has no leftover orbitals. sp3 allows single bonds only.