Understanding Valence Electrons and Shells in Periodic Table Elements
140 likes | 301 Views
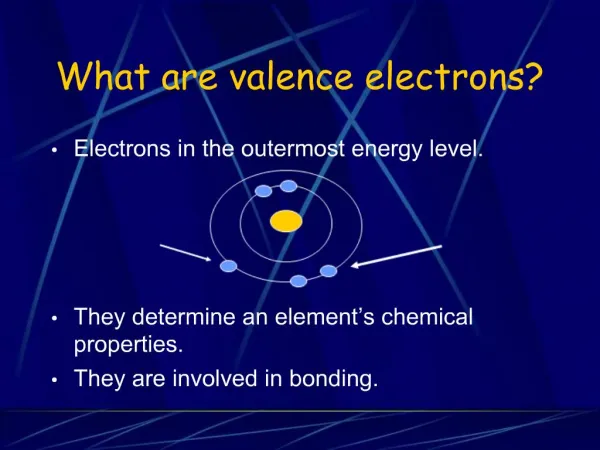
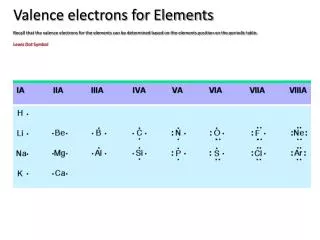
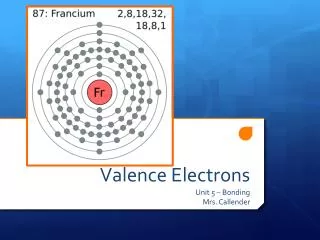
This guide explains the concept of valence electrons and electron shells in the periodic table. Each period indicates the number of electron shells an element has, while elements in a group share the same number of valence electrons. For example, Sodium (Na) has 3 shells and 1 valence electron, whereas Carbon (C) has 2 shells and 4 valence electrons. Learn how to determine these properties for various elements, and discover exceptions like Helium, which has only 2 electrons in its first shell but belongs to Group 8 due to its full outer shell.

Understanding Valence Electrons and Shells in Periodic Table Elements
E N D
Presentation Transcript
Periods • Each row is called a “period” • The elements in each period have the same number of electron shells 1st Period = 1 Shell 2nd Period = 2 Shells 3rd Period = 3 Shells 4th Period = 4 Shells www.chem4kids.com
Groups • Each element in a group has the same number of “valence electrons” Group 8 = 8 electrons Group 1 = 1 electron Group 2 = 2 electrons 3,4,5,6,7 Except He, it has 2 electrons
Transition Metals • Transition Metals have slightly different rules for shells and valence electrons. • This is something you will learn about in High School Chemistry. www.chem4kids.com
Determine the number of shells and the number of valence electrons for: Carbon - C 2nd Period = 2 shells 4th Group = 4 valence electrons
Determine the number of shells and the number of valence electrons for: Sodium - Na 3rd Period = 3 shells 1st Group = 1 valence electron www.chem4kids.com
10 Ne 20.18 PRACTICE? Name the element.__________ Number of protons?____ Average number of neutrons? ____ Number of shells ?____ Number of total electrons?____ Valence electrons ?_____
10 Ne 20.180 Answer? Name the element. Neon Number of protons? 10 p+ Number of neutrons? 10 n Number of shells ? 2 (2nd period) Number of total electrons? 10 e- Valence electrons ? 8 ve- (8th group)
4 Be 9.012 PRACTICE? Name the element.__________ Number of protons?____ Average number of neutrons? ____ Number of shells ?____ Number of total electrons?____ Valence electrons ?_____
4 Be 9.012 Answer? Name the element. Beryllium Number of protons? 4 p+ Number of neutrons? 5 n Number of shells ? 2 (2nd period) Number of total electrons? 4 e- Valence electrons ? 2ve- (2nd group)
16 S 32.096 PRACTICE? Name the element.__________ Number of protons?____ Average number of neutrons? ____ Number of shells ?____ Number of total electrons?____ Valence electrons ?_____
16 S 32.096 Answer? Name the element. Sulfur Number of protons? 16 p+ Number of neutrons? 16 n Number of shells ? 3 (3rd period) Number of total electrons? 16 e- Valence electrons ? 6ve- (6th group)
2 He 4.003 PRACTICE? Name the element.__________ Number of protons?____ Average number of neutrons? ____ Number of shells ?____ Number of total electrons?____ Valence electrons ?_____
2 He 4.003 Answer? • Helium is the exception in Group 8. • Since it has just one shell, that shell can only fit 2 electrons instead of 8. • It is in this group because all the elements have a full outer shell. Name the element. Helium Number of protons? 2 p+ Number of neutrons? 2 n Number of shells ? 1 (1st period) Number of total electrons? 2 e- Valence electrons ? 2ve- (8th group - why?)