Electrochemistry
Electrochemistry. CE 541. Electrochemistry is the relationship between Chemical Phenomena and Electrical Phenomena It is needed in Environmental Engineering to understand: Corrosion Electrochemical oxidation of wastes Analytical procedures Automatic monitoring of waste streams

Electrochemistry
E N D
Presentation Transcript
Electrochemistry CE 541
Electrochemistry is the relationship between Chemical Phenomena and Electrical Phenomena It is needed in Environmental Engineering to understand: • Corrosion • Electrochemical oxidation of wastes • Analytical procedures • Automatic monitoring of waste streams • Oxidation-reduction reactions
Current Flow in Solution Current can flow through: • Solution of electrolyte • Metallic conductors Characteristics of current flow through a metal: • Chemical properties of metal are not changed • Current is carried by electrons • Increase in temperature increases resistance
Characteristics of current flow through a solution: • Chemical change occurs in solution • Current is carried by ions • Increase in temperature decreases resistance • Resistance is normally greater than that with metals
Conductivity of Solution "Is its ability to carry an electrical current" Conductivity can be measured by a conductivity meter and it is affected by: • Number of ions • Type of ions E = IR Where: • E = electromotive force (volts) • I = current (amperes) • R = resistance (ohms)
And Where • l = length of conductor • A = cross-sectional area of conductor • = specific resistance of conductor (ohm-cm) • k = specific conductance (1 / ohm-cm) or siemens (S) Specific conductance is conductance afforded by 1 cm3 of an electrolyte solution
Conductivity cells are calibrated by determining the resistance of a standard solution (Rs) and the cell constant (C) can be found. C = ksRs In such cases 0.0100 N KCl is used in calibrating conductivity cells. For 0.0100 N KCl: ks = 0.00141185 S = 1411.8 S @ 25 C So, Specific Conductance of a Solution = C / R R needs to be determined
Equivalent Conductance () Where • N = normality of the solution • k = specific conductance or, and are equivalent ionic conductance of cations and anions, respectively
Table 3-3 shows equivalent ionic conductance @ 25 C in S-cm3/equivalent. Only ions can carry current. Un-ionized species of weak acids or bases will not carry current. Also uncharged soluble organics (ethanol and glucose) can not carry current. Study Example page 80
What is the approximate specific conductance at 25 C of a solution containing 100 mg/l of CaCl2 and 75 mg/l of Na2SO4
Current and Chemical Change Chemical change depends on: • Nature of solution (composition) • Nature of electrodes • Magnitude of electromotive force imposed
e- Cl- Cathode (-) Anode (+) H+ HCl Platinum Electrodes Applying a voltage of 1.3 vH2 evolves at CathodeCl2 evolves at Anode
To bring about 1 equivalent of chemical change at an electrode: • An Avogadro No of electrons must flow through the external circuit • This quantity of electrons is called the faraday (F) • The rate of flow of electrons gives the current (I) in amperes • 1 F is equivalent to an ampere of current flowing for 96500 seconds • An ampere is defined as a Coulomb per second • 1 F = 96500 Coulomb Study Example page 78
What weight of silver will pass into solution from a silver anode by the passage of 0.02 A of current through the solution for 24 hours?
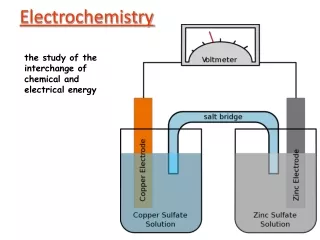
Electrochemical Cell • two electrodes will be connected by metallic conductor • electrons will flow • chemical change begins • electromotive force (emf) will be generated by the cell • this emf is a measure of the driving force of the chemical reaction occurring in the solution • the driving force represents the chemical potential or free energy of the reaction
Based on that, a relationship between electrical potential and chemical free energy can be found: Electrical Energy = EIt • E = emf in volts • I = current in amperes • t = time in seconds Electrical energy is expressed in Volt-Coulomb or Joule Electrical energy required to produce one mole of chemical change = zEF Where • z = number of electron-equivalent per mole • E = emf in volts • F = faraday or coulombs per equivalent
If reaction proceeds (E is +ve), then: where • G is the free energy • -zEF is the electrical energy
Consider the following reaction: aA + bB cC + dD Substituting in: we get:
The value of R in electrical units is: R = 8.314 J / K-mol At 25 C and converting ln to log
The emf can be found in Tables (Table 3-4) just like free-energy and enthalpy. The values in the table are for a reaction written for 1 mole of e- change, such as: If an electrochemical cell reaches the state of equilibrium, then: • no current can flow • emf is zero
In this case: since or log K = 16.9zE Study Examples page 85
Estimate the solubility-product constant for Mg(OH)2(s) at 25 C from standard electrode potential?
Chemical Kinetics Chemical kinetics deal with speed of reactions. If then, the rate of reaction could be: first – order reaction (exponent 1) • kCa second – order reaction (exponent 2) • kCa2 • kCaCb third – order reaction (exponent 3) • kCa3 • kCa2Cb • kCaCbCc • Ca, Cb, and Cc = concentrations of A, B, and C, respectively • k = rate constant
These are simple reaction rates, but in reality there are more complex equations. The unit of k depends on the reaction order and units of concentration of A, B, and C. The reaction rates are required in: • Microbial growth • Aeration • Disinfection • Radioactive decay
Zero-Order Reactions • They are independent of concentration • Most of biological growth occur in linear relationship over a range of concentrations of substance (C).
First-Order Reactions • The rate is directly proportional to the concentration • if we are dealing with a decay or decomposition reaction, then the rate can be expressed as • Unit of k is (1/time) and the –ve sign indicates the loss of material with time.
Integrating the above equation: Converting to log10
k = - slope of line [t versus ln(C/C0)] k = -slope 2.303 [t versus log10(C/C0)] Half-life (t1/2) In this case, t = t1/2 and C = (1/2)C0 Then Applications of 1st order reactions in Environmental Engineering: • Dissolution of gases in water • Removal of gases from water • Rate of death of microorganisms • Decomposition of organic matters (BOD5 test) Study Example page 89
The radioactive nuclide P32 has a half-life of 14.3 days. How long would a waste containing 10 mg/l of this nuclide have to be stored in order to reduce the concentration to 0.3 mg/l?
Second-Order Reactions The rate of reaction is proportional to the square of the concentration of one of the reactants or to the product of concentrations of two different reactants.
Ca and Cb are concentrations of A and B, respectively. Integrating (1) and (2), we obtain:
Consecutive Reactions If rates of reactions are 1st order, then:
Examples of consecutive reactions in Environmental Engineering
Enzyme Reactions Are used to describe the rate of biological waste treatment. The relationship between Substrate (S) and the rate of utilization per unit mass of enzyme or bacteria (V/E) Michaelis – Menton Relationship • Ef = free enzyme • S = substrate • EcS = enzyme-substrate complex Total enzyme concentration in the system = E = [Ef] + [EcS]
The rate of formation of enzyme-substrate complex is: The rate of complex formation rate of overall reaction. Therefore, d[EcS] / dt can be considered as ZERO when overall reaction rate is required to be determined. So:
Or The rate of product formation = overall rate of reaction Rate of product formation V = k[EcS]
Then Where • k is the maximum rate • Ks is the substrate concentration at arte = (1/2)k • Ks is called the "half velocity" constant V/E k'S when S<< Ks (1st order with respect to S) V/E k when S >> Ks (zero order with respect to S) Study Example page 95
Temperature Dependence of Reaction Rates "rates increase with increase in temperature" Rate doubles for each 10 C rise. Using Arrehenius equation: Where • T = temperature, K • R = universal gas constant • Ea = constant
Integrating k2 and k1 are rate constants at T2 and T1. In environmental engineering processes, the range of temperature is small. So T2T1 can be assumed constant.
Adsorption "sorption is the concentration or movement of contaminants from one place to another" "adsorption involves partitioning of contaminants from one phase to another" "adsorption is the process by which ions or molecules present in one phase tend to condense and concentrate on the surface of another phase"
Adsorption Physical • Weak • multi-layers • free moving • reversible Chemical • Strong • mono-layer • no movement • non-reversible in most cases Ion exchange • electrical attraction • smaller particles • have stronger attraction • trivalent have stronger attraction than monovalent ions
Activated Carbon • 1 gram has surface area of 1000 m2 • Pore size ranges between 10 to 1000 A • Adsorption of gas increases wit the increase I pressure Adsorption depends on: • Nature of materials • Concentration • Temperature
Freundlich Isotherm Freundlich found that: Where • q = mass of contaminant per unit weight of the adsorbent • C = concentration of solute after adsorption • K and n = constants (they should be evaluated for each soluble and temperature) Freundlich isotherm can be expressed as: