Atoms - Molecules - Ions
620 likes | 2.57k Views
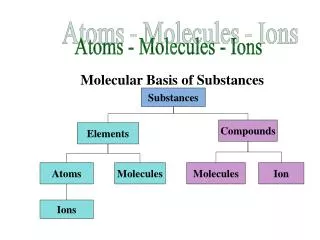
Atoms - Molecules - Ions. Molecular Basis of Substances. Substances. Compounds. Elements. Atoms. Molecules. Molecules. Ion. Ions. Periodic Table of the Elements. Hydrogen – 1 proton per atom. Carbon – 6 protons per atom. Elements.

Atoms - Molecules - Ions
E N D
Presentation Transcript
Atoms - Molecules - Ions Molecular Basis of Substances Substances Compounds Elements Atoms Molecules Molecules Ion Ions
Hydrogen – 1 proton per atom Carbon – 6 protons per atom Elements Elements contain one or more of the same type of atom! Examples include: Oxygen – 8 protons per atom Copper – 29 protons per atom Gold – 79 protons per atom
Why is the Periodic Table important to me? • The periodic table is the most useful tool to a chemist. • You get to use it on every test. • It organizes lots of information about all the known elements.
Pre-Periodic Table Chemistry … • …was a mess!!! • No organization of elements. • Imagine going to a grocery store with no organization!! • Difficult to find information. • Chemistry didn’t make sense.
Here are some of the symbols Dalton used for atoms of elements and molecules of compounds. He probably used a circle for each because, like the ancient Greeks, he thought of atoms as tiny, round hard spheres.
HOW HIS WORKED… Put elements in rows by increasing atomic weight. Put elements in columns by the way they reacted. SOME PROBLEMS… He left blank spaces for what he said were undiscovered elements. (Turned out he was right!) He broke the pattern of increasing atomic weight to keep similar reacting elements together. Dmitri Mendeleev: Father of the Table
Dmitri Medeleev gave us a functional scheme with which to classify elements. • Mendeleev’s scheme was based on chemical properties of the elements. • It was noticed that the chemical properties of elements increased in a periodic manner. • The periodicity of the elements was demonstrated by Medeleev when he used the table to predict to occurrence and chemical properties of elements which had not yet been discovered.
Mendeleev left blank spaces in his table when the properties of the elements above and below did not seem to match. The existence of unknown elements was predicted by Mendeleev on the basis of the blank spaces. When the unknown elements were discovered, it was found that Mendeleev had closely predicted the properties of the elements as well as their discovery.
The Current Periodic Table • Mendeleev wasn’t too far off. • Now the elements are put in rows by increasing ATOMIC NUMBER!! • The horizontal rows are called periods and are labeled from 1 to 7. • The vertical columns are called groups are labeled from 1 to 18.
Non-metals Periods Groups Transition metals Metalloids B,Si,Ge,As,Sb,Te Metals Periodic Table
Groups…Here’s Where the Periodic Table Gets Useful!! • Elements in the same group have similar chemical and physical properties!! • (Mendeleev did that on purpose.) • Why?? • They have the same number of valence electrons. • They will form the same kinds of ions.
Families on the Periodic Table • Columns are also grouped into families. • Families may be one column, or several columns put together. • Families have names rather than numbers. (Just like your family has a common last name.)
The Periodic Law • Similar physical and chemical properties recur periodically when the elements are listed in order of increasing atomic number.
Introduction • The periodic table is made up of rows of elements and columns. • An element is identified by its chemical symbol. • The number above the symbol is the atomic number • The number below the symbol is the rounded atomic weight of the element. • A row is called a period • A column is called a group
(A) Periods of the periodic table, and (B) groups of the periodic table.
Periodic Patterns • The chemical behavior of elements is determined by its electron configuration • Energy levels are quantized so roughly correspond to layers of electrons around the nucleus. • A shell is all the electrons with the same value of n. • n is a row in the periodic table. • Each period begins with a new outer electron shell
Each period ends with a completely filled outer shell that has the maximum number of electrons for that shell. • The number identifying the A families identifies the number of electrons in the outer shell, except helium • The outer shell electrons are responsible for chemical reactions. • Group A elements are called representative elements • Group B elements are called transition elements.
Chemical “Families” • IA are called alkali metals because the react with water to from an alkaline solution • Group IIA are called the alkali earth metals because they are reactive, but not as reactive as Group IA. • They are also soft metals like Earth. • Group VIIA are the halogens • These need only one electron to fill their outer shell • They are very reactive. • Group VIIIA are the noble gases as they have completely filled outer shells • They are almost non reactive.
Four chemical families of the periodic table: the alkali metals (IA), the alkaline earth metals (IIA), halogens (VII), and the noble gases (VIIIA).
Atoms - Molecules - Ions Metals – Nonmetals - Metalloids Metals: A material that has a characteristic luster or shine and that is a relatively good conductor of heat and electricity (Metal oxides react with water to form bases and are located on the left side of the table) Nonmetals: A element that does not exhibit the properties of a metal. (Nonmetal oxides react with water to form acids and are on the right side of the table) Metalloids: Elements with properties intermediate between those of metals and nonmetals.
Periodic Table: 1A Alkali metals Very reactive Metals never occur in nature as pure elements
Hydrogen • Hydrogen belongs to a family of its own. • Hydrogen is a diatomic, reactive gas. • Hydrogen was involved in the explosion of the Hindenberg. • Hydrogen is promising as an alternative fuel source for automobiles
Alkali Metals • 1st column on the periodic table (Group 1) not including hydrogen. • Very reactive metals, always combined with something else in nature (like in salt). • Soft enough to cut with a butter knife
Periodic Table: 2A Alkaline Earth metals
Alkaline Earth Metals • Second column on the periodic table. (Group 2) • Reactive metals that are always combined with nonmetals in nature. • Several of these elements are important mineral nutrients (such as Mg and Ca
Boron Family • Elements in group 13 • Aluminum metal was once rare and expensive, not a “disposable metal.”
Periodic Table: 4A allotropes of carbon
Carbon Family • Elements in group 14 • Contains elements important to life and computers. • Carbon is the basis for an entire branch of chemistry. • Silicon and Germanium are important semiconductors.
Nitrogen Family • Elements in group 15 • Nitrogen makes up over ¾ of the atmosphere. • Nitrogen and phosphorus are both important in living things. • Most of the world’s nitrogen is not available to living things. • The red stuff on the tip of matches is phosphorus.
Periodic Table: 6A Chalcogens
Oxygen Family or Chalcogens • Elements in group 16 • Oxygen is necessary for respiration. • Many things that stink, contain sulfur (rotten eggs, garlic, skunks,etc.)
Periodic Table: 7A Halogens Greek for “salt-forming” Very reactive
Halogens • Elements in group 17 • Very reactive, volatile, diatomic, nonmetals • Always found combined with other element in nature . • Used as disinfectants and to strengthen teeth.
Periodic Table: 8A Noble gases
The Noble Gases • Elements in group 18 • VERY unreactive, monatomic gases • Used in lighted “neon” signs • Used in blimps to fix the Hindenberg problem. • Have a full valence shell.
Transition Metals • Elements in groups 3-12 • Less reactive harder metals • Includes metals used in jewelry and construction. • Metals used “as metal.”
Names of Elements • The first 103 elements have internationally accepted names, which are derived from: • The compound or substance in which the element was discovered • An unusual or identifying property of the element • Places, cities, and countries • Famous scientists • Greek mythology • Astronomical objects.
What is a chemical symbol? • Chemical symbol tells us what atom it is • Always a CAPITAL LETTER, sometimes followed by a lower case letter • Ex: Oxygen O Nitrogen N Sodium Na • *Cobalt – Co *Carbon Monoxide – CO • (Co is an element, CO is a compound)
Chemical Symbols • There are about a dozen common elements that have s single capitalized letter for their symbol • The rest, that have permanent names have two letters. • the first is capitalized and the second is lower case. • Some elements have symbols from their Latin names. • Ten of the elements have symbols from their Latin or German names.
Charge (if ion) Atomic Mass Symbol Atomic Number
Hydrogen Protons: 1 Neutrons: 0 Electrons: 1 H 1 1
Sodium Protons: 11 Neutrons: 12 Electrons: 11 Na 23 11