SHOCK
750 likes | 1k Views
SHOCK. SHOCK. ADULT HEALTH II SUMMER, 2010. Jerry Carley MSN, MA, RN, CNE. What’s on your mind?????. Hopefully, Learning About S-H-O-C-K !. “….it’s just a urinary tract infection….”.

SHOCK
E N D
Presentation Transcript
SHOCK SHOCK ADULT HEALTH II SUMMER, 2010 Jerry Carley MSN, MA, RN, CNE
What’s on your mind????? Hopefully, Learning About S-H-O-C-K !
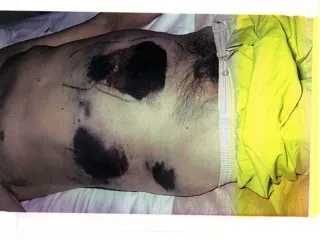
“….it’s just a urinary tract infection….” --December 30: Presented with complaint of dysuria---diagnosed as kidney stones & sent home on pain medications --Presented again on January 3rd in septic shock. --Pseudomonas aeruginosaurosepsis --Developed dissiminated intravascular coagulopathy (DIC) --partial gastrectomy (necrotic tissue) --January 23rd, hands and feet amputated --Comatose & on a ventilator *****Died January 23rd
Mariana Bridida Costa, January 2009:Headline: Brazilian Super Model Dies Mariana Bridida Costa, 20 y.o.
Questions To Ponder: Shock
1. Your client has multi-trauma & just arrived in the ER. The client’s urinary output is normal, whereas respiratory rate and heart rate are slightly elevated from baseline. Which of the following should the nurse suspect? • A. Early stage of shock • B. Compensatory stage of shock • C. Intermediate stage of shock • D. Refractory stage of shock
2. A client has been admitted with a gastrointestinal ulcer. The client is NPO and has a nasogastric tube in place connected to low intermittent suction. What form of shock should the nurse suspect / monitor this client for? • A. Distributive shock • B. Obstructive shock • C. Cardiogenic shock • D. Hypovolemic shock
3. A client in hypovolemic shock has been placed on a dopamine hydrochloride drip. Which parameter would indicate a desired client response to this drug? • A. Hypotension • B. Tachycardia • C. Increased cardiac output • D. Decreased mean arterial pressure
4. A nurse is monitoring a client who is receiving a dopamine hydrochloride drip for the treatment of shock. What symptom would indicate a possible overdose of this medication? • A. Pallor • B. Hypertension • C. Palmar erythema • D. Increased pulse deficit
5. What assessment is most appropriate for the client receiving sodium nitroprusside? • A. Assess for chest pain. • B. Assess blood pressure every 15 minutes. • C. Monitor urinary output every 30 minutes. • D. Observe the client’s extremities for color and perfusion.
6. Which manifestations should the nurse expect when caring for the client with distributive shock resulting from an anaphylactic event? • A. Increased heart rate and blood pressure • B. Increased blood pressure and cardiac output • C. Decreased blood pressure and respiratory rate • D. Decreased blood pressure and edema
7. The client has all the following clinical manifestations. Which one alerts the nurse to the probability of septic shock? • A. Hypotension • B. Pale, clammy skin • C. Anxiety and confusion • D. Oozing of blood at the IV site
End of Quiz(answers later) • 8. The average blood volume for an adult is 65 – 75 mL /Kg • The client weighs 209 lbs • What is the estimated blood volume for this patient?
At the conclusion of this presentation, the nurse will be able to: • 1. Define the concept known as SHOCK. • 2. Name, discuss, compare and contrast the four general categories of shock. • 3. Name, discuss, compare and contrast the four stages of shock. • 4. Name, discuss, compare and contrast etiology / risk factors associated with the four general categories of shock. • 5. Name the various diagnostic tests helpful in diagnosing and monitoring shock. • 6. Identify and Discuss nursing interventions associated with treatment of clients with shock • 7. Identify and Discuss the stages of shock; compare and contrast associated assessment findings with the stages of shock. • 8. Identify and discuss pharmacologic medications and interventions associated with the various types of shock.
C.O. = HR X SV • (CRAP) • contractility rate afterload preload • BP= C.O. X PVR • MAP= (2 x D) + S / 3
A Concept Map: Shock Hypovolemic Shock Insufficient oxygenation of tissues related to a sustained decrease in mean arterial pressure (MAP) NSG DX NURSING Interventions PATIENT Outcomes Hemodynamic Regulation Decreased Cardiac Output Cardiac / Pump Effectiveness Risk Factors Seth, 17 y.o. Multi-Trauma Cardiovascular: Tachycardia (+) Hypotension (+) Decreased PO2 (+) Respiratory: Tachypnea (+) Lungs CTA Integumentary: Pallor (+) Capillary Refill > 3 sec Kidney / Urinary: Output < 30 mL/hr SG > 1.035 CNS: Change in level of consciousness Pain, acute Glasgow= 10 Musculoskeletal: Deformity, ecchymosis and edema, both thighs Deformity, ecchymosis, crepitancebilat lower rib cage Shock Management *Chief Complaint: S/P MVC Pain Multiple Fractures Lower Extremities; Fractured lower two ribs, bilateral Medical History: Negative Medications: None Lab Studies: Hgb 12.2 Hct 55% WBC 18.8 BS 110 Physical Exam: HR 138 Resp 32 BP 88/68 Pulse Ox 85% Restless, Irritable F/E Management Risk for Ineffective Tissue Perfusion Circulation Status analyze IV Therapy Fluid Balance Deficient Fluid Volume Hypovolemia Management Pain Management Pain Immobilization / Pain Medications
A Tale of4 Patients Boyd, 17 y.o. S/P MVA, Multiple Trauma Obstructive Shock Hypovolemic Shock Roseline, 36 y.o. w/ hx Aortic Stenosis, CHF Frank 32 y.o. Anaphylaxis Septic Shock Distributive Shock Ian, 26 y.o. Meningococcal meningitis
Defined… • Shock: a state of inadequate tissue perfusion that impairs maintenance of normal cellular metabolism. Any condition that compromises oxygen delivery to tissues and organs can cause shock. STAGES:INITIAL COMPENSATORY PROGRESSIVE, REFRACTORY THE CAUSE OF SHOCK, CATEGORY OF SHOCK, AND STAGE OF SHOCK DIRECT THE SPECIFIC TREATMENT
Types of Shock(Classified by underlying cause) • Cardiogenic • Hypovolemic • Distributive • Obstructive “Pump Failure” or “Heart Failure” Decrease in intravascular volume of 10-15% or more Widespread vasodilation and capillary permeability (3 types…) (septic, neurogenic, anaphylactic) Mechanical blockage in the heart or great vessels
Progression of Shock (Stages) Initial No visible changes in client parameters, changes are now occurring on the cellular level only Compensatory Body is mounting measures to increase cardiac output to restore tissue perfusion and oxygenation. Progressive Compensatory mechanisms begin to fail Refractory IRREVERSIBLE; TOTAL BODY FAILURE
Risk Factors Pump failure due to myocardial infarction, heart failure, Cardiomyopathy, dysrhythmias, cardiac tamponade, valvular rupture or valvular stenosis • Cardiogenic • Hypovolemic • Distributive • Septic • Neurogenic • Anaphylactic • Obstructive Excessive fluid loss from diuresis, vomiting & diarrhea; Blood loss secondary to surgery, trauma, ob-gyn causes; Burns; Diabetic Ketoacidosis Endotoxins and other mediators causing massive vasodilation. Most common is gram-negative bacteria. Loss of sympathetic tone causing massive vasodilation. Trauma, spinal shock, and epidural anesthesia are among the causes. Antigen-antibody reaction causing massive vasodilation. Blockage of great vessels. Cardiac valve stenosis, pulmonary Embolism, and aortic dissection are among the causes.
Diagnostic Procedures • ABG’s • Hemodynamic Monitoring • Cardiogenic Shock: • EKG, Echocardiogram, CT Scans, Cardiac catheterization, CXR, Cardiac Enzymes • Hypovolemic Shock: • Hgb & Hct; Type & Crossmatch; investigate for source of bleeding • Septic Shock: • Cultures: blood, urine, wound • Coagulation tests: PT, PTT, INR • Obstructive Shock: • Echocardiogram, CT scan, • Monitor Signs & Symptoms • Hypoxia • Hypotension (MAP < 60 mm Hg) • Tachycardia, weak thready pulse
Stages of Shock With Associated Assessment Changes -Miscellaneous: May see rashes with septic or anaphylactic shock. -May see angioedema with anaphylactic shock. -Rales (coarse crackles) are possible with cardiogenic shock. -SEIZURES MAY OCCUR WITH ALL FORMS OF SHOCK. -FEVER MAY OCCUR WITH ALL FORMS OF SHOCK— BUT ESPECIALLY SEPTIC SHOCK
Shock Position Evidence-Based Practice Update: Friedrich Trendelenberg
Assess & Monitor OXYGENATION • PRIORITY: ______________ • Vital signs • Urinary Output • LOC • Cardiac Rhythm • Skin color, temperature, moisture, capillary refill, turgor • Symptoms related to system: • chest pain, change in heart sounds, lung sounds, bowel sounds, neurological status
NANDA’s • Decreased Cardiac Output • Impaired Gas Exchange • Ineffective Tissue Perfusion • Deficient Fluid Volume • Anxiety
Cardiogenic Shock • When thinking about the hemodynamics of cardiogenic shock, keep it simple: • The components of cardiac output are: Contractility, Rate, Afterload, and Preload, or “CRAP.” To manage these patients, you’ve got to know CRAP! (This acronym has long been passed down to many a critical care and cath lab staff and is helpful when managing cardiogenic shock). • Every therapeutic intervention is aimed towards improving or altering a component of cardiac output — or something in CRAP.
MAP Mean Arterial Pressure • MAP = [(2*D)+S] / 3 • [ (2 x 60) + 100] / 3 = 73.3 mm Hg • It is believed that a MAP that is greater than 60 mmHg is enough to sustain the organs of the average person. • If the MAP falls significantly below this number for an appreciable time, the end organ will not get enough blood flow, and will become ischemic. PT’s BP = 100/60 mm Hg
Hypovolemic Shock BACK ! • Average Blood Volume = 65-75 mL /kg • 75 ml/Kg X 75 kg = 5625 mL = 5.625 L EBL 10% - 15% Yields Hypovolemic Shock 0.10 x 5625 = 560 mL 0.15 x 5625 = 844 ml 5-6 Liters
Shock Pharmacology: “Pressor Agents”
Milrinone / Amrinone • Belong to new class of agents “Bipyridines” • Non-receptor mediated activity based on selective inhibition of Phosphodiesterase Type III enzyme resulting in cAMP accumulation in myocardium • cAMP increases force of contraction and rate and extent of relaxation of myocardium • Inotropic, vasodilator and lusotropic effect • ( lusotropic= direct improvement of the relaxation phase of the LV.)
Amrinone(Inacor®) • First generation agent - limited use now • Long half-life (4.4 hours) with potential for prolonged hypotension after loading dose • Associated with thrombocytopenia • Dosage: Load with 0.75 mg/kg with infusion rate of 5-10 mcg/kg/min • Milrinone is preferred drug from this group
Milrinone(Primacor®) • Increases CO by improving contractility, decreased SVR, PVR , lusotropic effect; decreased preload due to vasodilatation • Unique in beneficial effects on RV function • Half-life is 1-2 hours • Load with 50 mcg/kg over 30 mins followed by 0.3 to 0.75 mcg/kg/min • No increase in myocardial O2 requirement
Epinephrine • Actions are dose dependent (mcg/kg/min): • 0.02-0.08 = mostly beta1 and beta2 stimulation. • increased cardiac output • mild vasodilation • 0.1-2.0 = mix of beta1 andalpha1 • increase cardiac output • increase SVR = vasoconstriction • > 2.0 = mostly alpha1 • increase SVR, and may decrease CO by increasing afterload
Dopamine • Intermediate product in the enzymatic pathway leading to the production of norepinephrine; thus, it indirectly acts by releasing norepinephrine. • Directly has alpha, beta and dopaminergic actions which are dose-dependent. • Indications are based on the adrenergic actions desired.
Dopamine • Improve renal perfusion 2-5 mcg/kg/min • Improve C.O. in mild to moderate Cardiogenic or Distributive Shock 5-10mcg/kg/min • Post-resuscitation stabilization in patients with hypotension (in conjunction with fluid therapy) 10-20mcg/kg/min
Venodilators / Vasodilators Classified by site of action -Venodilators: reduce preload - Nitroglycerin -Arteriolar dilators: reduce afterload Minoxidil and Hydralazine -Combined: act on both arterial and venous beds and reduce both pre- and afterload Sodium Nitroprusside (Nipride)
Nipride ®nitroprusside -Vasodilator that acts directly on arterial and venous vascular smooth muscle. -Indicated in hypertension and low cardiac output states with increased SVR. -Also used in post-operative cardiac surgery to decrease afterload on an injured heart. -Action is immediate; half-life is short; titratable action.
Nipride ® -Toxicity is with cyanide, one of the metabolites of the breakdown of nipride. -Severe, unexplained metabolic acidosis might suggest cyanide toxicity. -Dose starts at 0.5 mcg/kg/min and titrate to 5 mcg/kg/min to desired effect. May go higher (up to 10 mcg/kg/min) for short periods of time.
Nitroglycerin • Direct vasodilator as well, but the major effect is as a venodilator with lesser effect on arterioles. • Not as effective as nitroprusside in lowering blood pressure. • Another potential benefit is relaxation of the coronary arteries, thus improving myocardial regional blood flow and myocardial oxygen demand.
NTG -Used to improve myocardial perfusion following cardiac surgery -Dose ranges from 0.5 to 8 mcg/kg/min. Typical dose is 2 mcg/kg/min for 24 to 48 hours post-operatively -Methemoglobinemia is potential side effect
Isoproterenol(Isuprel ®) -Synthetic catecholamine -Non-specific beta agonist with minimal alpha-adrenergic effects. -Causes inotropy, chronotropy, and systemic and pulmonary vasodilatation. -Indications: bradycardia, decreased cardiac output, bronchospasm (bronchodilator). -No longer available in some markets
Isoproterenol -Occasionally used to maintain heart rate following heart transplantation. -Dose starts at 0.01 mcg/kg/min and is increased to 1.0 mcg/kg/min for desired effect.
Selecting inotropic and vasopressor agents for specific hemodynamic disturbances BP or SVR>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>> Hemodynamic pattern
Cardiogenic Shock and Hemodynamic Support: A Realistic Management Approach- Mary Dahling, RN, MSN, CCRN, CNS, Cardiothoracic Surgery, Sentara Norfolk Hospital, Norfolk, Virginia What is cardiogenic shock? • By definition, cardiogenic shock is decreased cardiac output and evidence of tissue hypoperfusion in the presence of adequate intravascular volume.1 The presence of adequate intravascular volume is important. This differentiates cardiogenic shock from other types of shock, which typically have a relative or an absolute volume deficit. With cardiogenic shock, the patient usually has enough intravascular volume — it’s just not going to the right place(s) due to pump failure. These patients present with sustained hypotension defined by blood pressure less than 80 mmHg (or 90 mmHg if on pressors, inotropic agents or intraaortic balloon pump support) for greater than 30–60 minutes, a cardiac index under 1.8 liters/ minute, in the presence of a left ventricular end-diastolic pressure (LVEDP) or pulmonary capillary wedge pressure (PCWP) greater than or equal to 18 mmHg.4,5 When dealing with “cardiogenic shock,” think “decreased forward flow” because interventions must be aimed toward restoration of forward volume flow. We lose if we don’t perfuse! Cath Lab Digest - ISSN: 1073-2667 - Volume 11 - Issue 11 - November 2003 - Pages: 20 - 25