SON
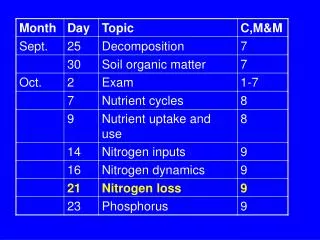
NH 3 +. N 2. NO x , N 2 O, N 2. Litter. SON. DON. NH 4 +. NO 3 -. Leaching. Erosion. Leaching. When is NH 3 produced?. NH 4 + + OH -. NH 3 + H 2 O. If soil pH >9.3, then >50% of NH 4 + goes to NH 3. Ammonia Volatilization

SON
E N D
Presentation Transcript
NH3+ N2 NOx, N2O, N2 Litter SON DON NH4+ NO3- Leaching Erosion Leaching
When is NH3 produced? NH4+ + OH- NH3 + H2O If soil pH >9.3, then >50% of NH4+ goes to NH3 • Ammonia Volatilization • NH3 (ammonia gas) is emitted due to the pH-dependent equilibrium between NH4+ and NH3 What controls the production of NH3? • Depends on pH and [NH4+] What types of ecosystems show lots of ammonia volatilization?
NH3+ N2 NOx, N2O, N2 Litter SON DON NH4+ NO3- Leaching Erosion Leaching
When are NO, N2O, and N2 produced? Nitrification – NO, N2O Denitrification – NO, N2O, N2
NO3- N2 N2O NO2- NOx • Denitrification--reduction of nitrite and nitrate • NO2- and NO3- reduced to NOx, N2O, and N2 • Facultative anaerobic bacteria that use NO2- or NO3- as an alternate electron acceptor when O2 is low; get C from organic matter • Rate depends on [O2], [NO3-], C-availability What types of ecosystems show lots of denitrification?
Denitrification--Fate • Gaseous NOx, N2O, and N2 can be scavenged by microbes and plants, or may be lost from the ecosystem to the atmosphere
What controls the production of NO and N2O? • During Nitrification • Rate: as flow increases, “leaks” increase • 10 to 20 times as much NO is produced as N2O • During Denitrification • Rate: as flow increases, “leaks” increase • When NO3- is relatively more abundant than labile organic carbon, more N2O is produced than N2 • N2O > N2 in conditions of low pH, low temp., and low O2 • Most NO gets reduced
What controls how much NO and N2O leaves the ecosystem? • Rate of production • Soil texture: diffusion path length to escape soil • Stratification of microbial activity • Plant canopy cover
N2 is the major pathway for return of N from ecosystems to the atmosphere • After N2, loss of NOX = NH3 >> N2O
If N trace gasses are << 10% of N losses from most ecosystems, why do we care? Because they have large effects on atmospheric chemistry Global emissions: 23% of NOx and 61% of N2O comes from soil The global atmospheric N budget is unbalanced
In the atmosphere… • Ammonia—NH3 • Interacts with acids in troposphere • NH3 + H2SO4 (NH4)2SO4 • Reduces acidity of rain • Deposited downwind
In the atmosphere… • Nitric Oxide—NOx • Highly reactive in chemistry of the troposphere • Lifetime of <1 day, depending on sunlight... • Lifetime of <1 day, depending on sunlight... • Regulates OH concentration, which determines “scrubbing capacity” of the atmosphere • Regulates concentrations of CO, CH4+ and other hydrocarbons • Regulates the photochemical production of “bad” ozone • Suffers oxidation to HNO3, a component of acid rain
In the atmosphere… • Nitrous Oxide—N2O • Stable, residence time in atmosphere of 150 years • Migrates to stratosphere: • Radiatively active: 150 x more thermal adsorption capacity than CO2 • Undergoes photolysis to NO in stratosphere, resulting in destruction of “good” ozone
Human activities increase gaseous losses of N • Intensification of agriculture • Livestock production Can we manage systems to maintain economic gains and reduce N losses?
Farmer’s fields Alternative practice = fertilizer application Matson et al 1998
Fertilizer application after plant establishment • Reduced gas emissions by 50% • Did not affect grain yield • Increased farmer’s profits 20%
NH3+ N2 NOx, N2O, N2 Litter SON DON NH4+ NO3- Leaching Erosion Leaching
Nitrogen leaching losses • DON is the dominant form of loss in most undisturbed systems
Temperate forests in Chile and Argentina receive little or no N inputs from anthropogenic sources Perakis and Hedin 2002
Nitrogen leaching losses • DON is the dominant form of loss in most undisturbed systems • NO3- leaching losses dominate • In disturbed systems where plant uptake is reduced • In systems where N is in excess of plant demand, such as agricultural or weathered tropical soils, or sites that receive high deposition inputs
When NO3- leaches out... • Removes a cation (Ca+, K+, NH4+) to balance solution charge • When cations are depleted, NO3- removes H+ or Al 3+