lipids
lipids. www.themegallery.com. By Dr. Batoul Izzularab. 3. 4. Simple lipids. Triglycerides Triglyceride is a neutral fat are ester of fatty acids with glycerol. . Simple lipids.

lipids
E N D
Presentation Transcript
lipids www.themegallery.com
Simple lipids • Triglycerides • Triglyceride is a neutral fat are ester of fatty acids with glycerol.
Simple lipids • Neutral fats are oil and butter which are liquid or solid at room temperature due to the presence of unsaturated and saturated fatty acids respectively • Neutral fats are insoluble in water, soluble in fat solvents • Neutral fats react with alkalies (NAOH) producing soap and glycerol this process known as saponification.
Simple lipids • Neutral fats are hydrolyzed by pancreatic lipase into glycerol and fatty acid.
Simple lipids: • Vegetable oils (cotton seed oil, sun flower oil and olive oil) that contain unsaturated fatty acids, the latter can be saturated (hydrogenated) in the prencses of metal as acatalyst i.e. nickel). This process is used in manufacture of margarine.
Simple lipids • Oils can be saturated with iodine at a position of double bond this reaction indicates the degree of unsaturation.
Simple lipids • Rancidity: means change in odour and taste of oil and fats when stored for long period and exposed to oxygen, moisture and bacteria. There are two type of Rancidity: • Hydrolytic rancidity: this happens by bacterial action, that hydrolysis fats and oils into glycerol and free fatty acids • Oxidative rancidity: oxidation occurs at the sites of double bond, leading to formation of peroxides fatty acids aldhydes, dicarboxylic acids and free radicals
Simple lipids • How to prevent the occurrence of rancidity • Storage of fats and oils in dry space i.e. no moisture and no bacteria • Fill the container to its rim to avoid exposure to oxygen • Addition of antioxidant which prevent peroxide formation Also simple lipids are Naturally occurring antioxidants: Vitamin E, Vitamin C, urate, B carotene, peroxide des mutase.
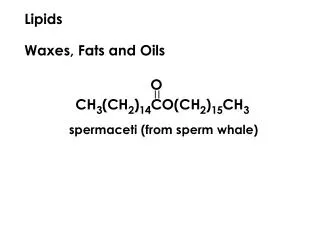
Waxes • Waxes are simple lipids and esters of fatty acides with higher monohydric alcohol (e.g. actyl alcohol) Bee wax is an example • Waxes are important in the manufacture of lubricant, polishes, ointment and cosmetics. • Waxes are not utilized by human as nutrient
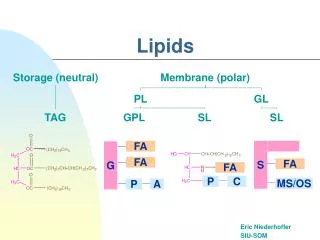
Complex or conjugated lipids Complex lipids are group of lipids on hydrolysis give rise to fatty acids, glycerol (may be other alchol) and in addation non lipids group name prosthetic group. According to this group complex lipids are classified into Phospholipids Phospholipids are classified according to alcohol present into Glycerophospholipids :( which contain glycerol) Sphinogophospholipids :( which contain sphingosine)
Glycerophospholipids • Phosphotidic acids= glycerol +2 F.AS+ phosphoric acid • Phosphatidic acids is present in a small amount in tissues • It is the mother material of other glycerophospholipids
Lecithin (phosphotidyl choline) • Phosphatic acid +choline • Widely distributed in tissue especially in plasma cell membrane
Cephalins ( phosphatid, ethanolamine) • Phosphatidic acids ethanolamine present in brain and nerve tissues.
Phosphatidyl serine: Phosphatidic acid + serine present in cell membrane and nerve tissue
Phosphatidyl inositol • Phosphatidic acid + Inositol • Phosphotidyl insitol 4, 5 biphosphate is an important constituent of cell membrane phospholipids upon specific stimulation (hormonal or chemical) cleavage of phosphotidyl insitol 4,5 bisphosphate into insitol triphosphate and diacylglycerol the later two acts as signal or second messenger
Sphinogophospholipids • Sphingophospholipids are group of phospholipids that contain sphingosine base instead of glycerol e.g. sphingomyelin that formed of • Sphingosine • Fatty acids molecules • Phosphoric acids • Choline • The combination of sphingosine and fatty acids is known are ceramide
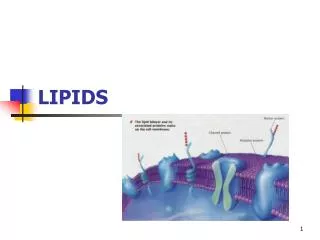
Biomedicalimportanceofphospholipids • Phospholipids are important constituent of all cells , membrane , mitochondria and golgi body • Phospholipids are important constituent of brain and nerve tissue • Phospholipids play important role in the process of lipids absorption in the intestine • Phospholipids are necessary in the process of lipids transport in the plasma to liver to subcutaneous tissue and to adipose tissue for the storage • Phospholipids play important role in mobilization of fat from the liver preventing and curringfatty liver
Glycolipids • Glycolipids are conjugated lipids in which the prothetic group is carbohydrate, glucose and galactose • Glycollipids chemically are ceramide (sphingosine + fatty acids) and one or more sugar (glucose or galactose) • Galactosylceramide: present in nerve and brain tissues • Glucosylceramide: present in outer leaflet of plasma membrane
Lipoprotein There conjugated lipid in the prosthetic group is protein the presence of protein mol. Attached to lipids play important role in transport of lipids (hydrophobic) in the watery plasma , otherwise obstruction of blood vessels and capillaries. According to ratio between lipid and protein content present in lipoprotein mol, lipoprotein are classified The density decreased when there is a high lipid content in relation to protein and vice versa Lipoprotein are classified into
Chylomierons Chylomierons are lipoprotein particles contain high amount of TAG, phospholipids and small amount of protein named apoprotien B- 48 The important of chylomicron is the transport of digested lipids from alimenteary canal to blood plasma
Very low density lipoprotein (VLDL) VLDL are conjugated lipids contain very small amount of protein named apoprotien 100 VLDL are synthesized in liver cells VLDL function in transport at of fats and cholesterol from liver to blood Deficiency of VLDL syntheses lead to deposition of fats in liver consequently fatty liver occurrence
Low density lipoprotein LDL This lipoproptien formed in the blood in the course of VLDL break down. High elevated LDL blood level carry risk of hyperlipidemia, hypercholesterolemia and atheroseclsis
High density lipoprotein HDL HDL syntheses by intestinal cell mucosa and liver cell then transported to the blood HDL contain great amount of protein in relation to lipids The apoprotien present in HDL are Apo A-1apo E II and Apo D HDL play important role in reverse transport of cholesterol i.e. from tissues and blood to liver to be excreted HDL guard against hypercholesterolemia
Derived lipids These are substance derived from hydrolysis of neutral fats, oil and butter into fatty acids saturated or unsaturated) and glycerol and also the associated compounds as: Sterol (cholesterol, bile salt and vitD3) Steroid (male & female sex hormone and Super-renal cortical hormone Fat soluble vitamins Keton bodies
Sterol Cholesterol It is derived lipids present only as animal source cholesterol is widely distributed in tissue e.g brain, nerve suprarenal cortex kidney and carpusleuteum Cholesterol occurs in the blood either free or conjugated form normally its level is 150-250 mg% cholesterol is the presursose of vit D3 and steroid hormones and also can be converted into bile salt Cholesterol is excreta in billiary secretion Cholesterol can be precipitated in gall stones or can be precipitated in the intima of the blood vessels leading to atherosclerosis, hypertension and coronary heart disease
Steroids • Super-renal cortical hormone • Glucocorticoid • Controls carbohydrate metabolism • Has anti-inflammatory effect • Has immune suppression effect • Mineralocorticoids • It increases reabsorption of Na+, Cl-, and water in proximal convoluted tubule of the kidney. increase K+ excretion
Thank You! www.themegallery.com