The Octet Rule
The Octet Rule. Noble gases are unlike any other group of elements on the periodic table because of their extreme stability. Each noble gas has eight valence electrons , except for helium, which has two.

The Octet Rule
E N D
Presentation Transcript
The Octet Rule • Noble gases are unlike any other group of elements on the periodic table because of their extreme stability. • Each noble gas has eight valence electrons, except for helium, which has two. • The modern model of how atoms react to form compounds is based on the fact that the stability of a noble gas results from the arrangement of its valence electrons.
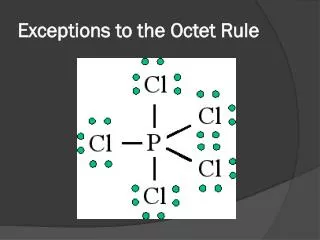
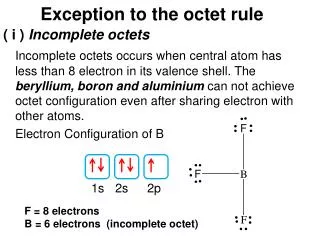
The Octet Rule • This model of chemical stability is called the octet rule. • The octet rule says that atoms can become stable by having eight electrons in their outer energy level, as shown in the noble gas, Neon, (or two electrons in the case of some of the smallest atoms). • Elements become stable by achieving the same configuration of valence electrons as one of the noble gases, a noble gas configuration.
Electrons Can Be Transferred • Sodium is in Group 1, so it has one valence electron. • Chlorine is in Group 17 and has seven valence electrons. • How can the valence electrons of atoms rearrange to give each atom a stable configuration of valence electrons? • If the one valence electron of sodium is transferred to the chlorine atom, chlorine becomes stable with an octet of electrons. • Because the chlorine atom now has an extra electron, it has a negative charge. • Also, because sodium lost an electron, it now has an unbalanced proton in the nucleus and therefore has a positive charge.
Electrons Can Be Transferred • Now that each atom has an octet of outer-level electrons, they are no longer neutral atoms; they are charged particles called ions. • An ion is an atom or group of combined atoms that has a charge because of the loss or gain of electrons. • ionic compound : a compound that is composed of ions. • Note that only the arrangement of electrons has changed. Nothing about the atom’s nucleus has changed.
Formation of an Ionic Compound • Remember that objects with opposite charges attract each other. • The strong attractive force between ions of opposite charge is called an ionic bond. • ionic compound : isthe force of the ionic bond holds ions together . • Unprotected aluminum metal reacts with oxygen in air, forming the white coating you can observe on aluminum objects such as lawn furniture.
Explaining the Properties of Ionic Compounds • Ionic compounds are composed of well-organized, tightly bound ions. • These ions form a strong, three-dimensional crystal structure. • Ionic compounds are crystalline solids at room temperature. • Ionic compounds usually have to be heated to high temperatures in order to melt them because the attractions between ions of opposite charge are strong.
Electrolytes • Another physical property of ionic compounds is their tendency to dissolve in water and conduct electricity in the liquid (melted) state. • Any compound that conducts electricity when melted or dissolved in water is an electrolyte. • In order to conduct electricity, ions must be free to move because they must take on or give up electrons.
Formation of Water by Electron Sharing • The stability of the atoms in a water molecule results from a cooperative arrangement in which the eight valence electrons (six from oxygen and one each from two hydrogens) are distributed among the three atoms.
Electron Sharing Produces Molecules • covalent bond: is the attraction of two atoms for a shared pair of electrons . • Notice that in a covalent bond, atoms share electrons and neither atom has an ionic charge. • covalent compound: is a compound whose atoms are held together by covalent bonds. • Water is a covalent compound. • A moleculeis an uncharged group of two or more atoms held together by covalent bonds. • Ethanol, also known as ethyl alcohol, is a typical covalent compound.