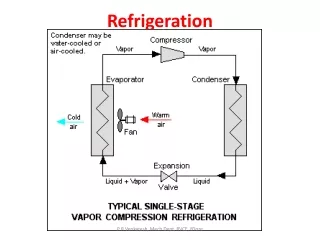
Refrigeration
Refrigeration. Plants – combine carbon dioxide from atmosphere, moisture and traces of chemicals from the soil to form all constituents they need. Energy from sunlight and green parts of plants can effect this synthesis.

Refrigeration
E N D
Presentation Transcript
Refrigeration Plants – combine carbon dioxide from atmosphere, moisture and traces of chemicals from the soil to form all constituents they need. Energy from sunlight and green parts of plants can effect this synthesis.
Animals cannot do this They have to get food from plants or other animals. From their food they absorb what they require in their digestive system and reject the rest. Many micro-organisms live on the same food as men. Our battle is to get the food before the micro-organisms.
In this battle the micro-organisms are well placed – size so small they are ever present in the atmosphere. Any food exposed to atmosphere at ordinary temperature – appropriate organisms will start to grow. They consume the food like animal – absorbing what they like and rejecting the remainder. Rejected remainder is often toxic – leading to food poisoning.
Micro-organisms can be divided in three groups – moulds, yeast and bacteria. • Moulds – start microscopic – can develop into clearly visible growths. • Yeast – reproduce into microscopic individuals – presence known by the alcohol their digestive process rejects – leading to fermentation. • Bacteria - also multiply as microscopic individuals- their waste products often giving rise to evil smells of putrefaction as well as being toxic.
Each type of micro-organisms has its own environment of temperature, atmosphere (gaseous composition and humidity) and food for optimum growth. Water is required by everyone. Hence drying can also lead to food preservation. • Control of micro-organisms by subjecting them to low temp. is basis of refrigerated carriage of foodstuffs.
There are three main temperature group of micro-organisms – • 36.70 C for optimal growth. This group includes bacteria. They practically cease growth at 00 C. • Optimal growth at 210 C, which are capable of rapid growth at 00 C and which will grow on unfrozen substances below this temperature. This group includes yeast and moulds and bacteria. • Micro-organisms, mainly yeasts and moulds, which will grow on frozen substances down to about -70 C. • Below –80 C no growth or reproduction of micro-organisms occurs.
For each living process there is an optimum temp. at which the process occurs at the maximum rate. As temp. falls below the optimum, the rate of living falls off until at low enough temperatures life ceases. As temperature rises above the optimum, different chemical processes take over from the normal living reactions until at high enough temperature life again ceases . Optimum temperature varies for each life process, but all lie within the range of –80 C to 1000 C, outside which life is virtually unknown.
Dead animal products • With dead animal produce, the primary purpose of refrigeration is to delay or prevent the developments of micro-organisms (moulds, yeast and bacteria) which live on the produce. Cold storage also retards the slow chemical changes, such as the oxidation of fats, which whilst not rendering the produce unfit for human consumption do adversely affect flavours.
Dead animal products are carried as frozen cargo below –80 C. None of the micro-organisms that cause decay and putrefaction grow and multiply below this temperature, although some may survive in dormant state to resume growth if temperature is raised subsequently. Further temperature reduction is required to slow chemical changes that impair flavour. For beef and lamb –100 C is adequate. For pork –12 to –150 C. Poultry and fish –180 C is desirable.
Fruits and vegetables • The living process continues after picking of fruits and vegetables. After picking at immature stage the ripening process continues until it is fully ripe and finally overripe. Cool storage delays the ripening process. Second benefit is delaying of onset of mouldiness as they are likely to be attacked by moulds. Hence by refrigeration, we keep it alive and let it ripen slowly. The water content of fruits and vegetables is 80-90%. If brought below freezing point, changes in physical structure will take place and will be killed. They are to be carried in chilled condition above their freezing point.
Common between fruit and animal produce –provide conditions unsuitable for growth of organisms which live on the produce. Chemical reaction proceed more slowly at low temperature. 50C drop in temperature halves the rate of reaction.
Units of Refrigeration Capacity • Tonnage – One Ton of Refrigeration implies latent heat of one tonne of ice or the heat required to convert one tonne of water to ice. Equivalent to 12,000 BTUs. One BTU is the heat required to raise one pound of water by one degree Fahrenheit. • One calorie is the heat required to raise one gram of water by one degree Celsius. • Joule is the mechanical equivalent of heat, work done when one a force of one Newton moves through one metre. • One Joule = 4.2 calories • Calculate the conversion factor from BTUs to Kilocalories
Refrigerant • Refrigeration depends upon a substance called the refrigerant, which can readily be converted from liquid into a vapour (evaporation) and also from a vapour into a liquid (condensation) within a narrow range of pressures. Refrigerants are those fluids, which are used as working fluids, for example in vapour compression refrigeration systems. These fluids provide refrigeration by undergoing a phase change process in the evaporator. • A refrigerant gives up heat by condensing at high temperatures and pressures and absorbs heat by evaporating at low temperatures and pressures.
REFRIGERANTS Halocarbon based Non-Halocarbon based Ethane based Methane based CFCs HCFCs HFCs Primary Secondary Pure Blended Azeotropic Near Azeotropic Zeotropic
Azeotropic Refrigerants: Mixture of two or more refrigerants with similar boiling points. Where the change of phase takes place at a specific single temperature.ex: R-500;R-502; R-503; R-507. They can be charged as a liquid or vapour. • Zeotropic Refrigerants: Mixture of two or more refrigerants with dissimilar boiling points. Where the change of phase takes place over a temperature range of over 5.56 degrees C (10 deg F).ex: R-404a; 407c. They should be charged as a liquid. • Near Azeotropic Refrigerants: : Mixture of two or more refrigerants with dissimilar boiling points, but the change of phase takes place over a temperature range of less than 5.56 degrees C (10 deg F). R-410a. They should be charged as a liquid. • TEMPERATURE GLIDE is the range of temperature over which change of phase occurs. Important because fractionating of refrigerant occurs especially when there is a leak. Also complicates the charging process.
Generation of Refrigerants • First Generation 1830-1930s ex: CO2; NH3; HCs;SO2 etc. usefulness of volatile compounds • Second Generation: 1931-1990s ex: CFCs; HCFCs; safety & durability • Third Generation; 1990-2010s ex: HCFCs and HFCs; Ozone Layer protection • Fourth Generation; 2010 onwards ex: Pure & Blended HCs; Global warming; high efficiency
Refrigerant selection criteria • Selection of refrigerant for a particular application is based on the following requirements: • Thermodynamic and thermo-physical properties • Environmental and safety properties, and • Economics
Thermodynamic and thermo-physical properties • The requirements are: • Suction pressure: At a given evaporator temperature, the saturation pressure should be above atmospheric for prevention of air or moisture ingress into the system and ease of leak detection. Higher suction pressure is better as it leads to smaller compressor displacement • Discharge pressure: At a given condenser temperature, the discharge pressure should be as small as possible to allow light-weight construction of compressor, condenser etc.
3. Pressure ratio: Should be as small as possible for high volumetric efficiency and low power consumption 4. Latent heat of vaporization: Should be as large as possible so that the required mass flow rate per unit cooling capacity will be small 5. Isentropic index of compression: Should be as small as possible so that the temperature rise during compression will be small
Liquid specific heat: Should be small so that degree of subcooling will be large leading to smaller amount of flash gas at evaporator inlet • Vapour specific heat: Should be large so that the degree of superheating will be small • Thermal conductivity: Thermal conductivity in both liquid as well as vapour phase should be high for higher heat transfer coefficients
Viscosity: Viscosity should be small in both liquid and vapour phases for smaller frictional pressure drops • The freezing point of the refrigerant should be lower than the lowest operating temperature of the cycle to prevent blockage of refrigerant pipelines. • High critical temperature
Environmental properties • The important environmental properties are: • Ozone Depletion Potential (ODP) • Global Warming Potential (GWP) • Total Equivalent Warming Index (TEWI) • Atmospheric Lifetime • Chlorine Leading Potential
Safety and Other Properties • TLV (Threshold Limit Value) • Toxicity • Flammability • Chemical stability • Compatibility with common materials • Miscibility with lubricating oils • Dielectric strength • Ease of leak detection
Ozone Depletion Potential (ODP): According to the Montreal protocol, the ODP of refrigerants should be zero, i.e., they should be non-ozone depleting substances. Refrigerants having non-zero ODP have either already been phased-out (e.g. R 11, R 12) or will be phased-out in near-future (e.g. R22). Since ODP depends mainly on the presence of chlorine or bromine in the molecules, refrigerants having either chlorine (i.e., CFCs and HCFCs) or bromine cannot be used under the new regulations
Global Warming Potential (GWP) Refrigerants should have as low a GWP value as possible to minimize the problem of global warming. Refrigerants with zero ODP but a high value of GWP (e.g. R134a) are likely to be regulated in future.
Total Equivalent Warming Index (TEWI) • The factor TEWI considers both direct (due to release into atmosphere) and indirect (through energy consumption) contributions of refrigerants to global warming. Naturally, refrigerants with as a low a value of TEWI are preferable from global warming point of view.
Atmospheric Lifetime • HFC125, the major component of HFC blend refrigerants, has an atmospheric life of 29 years, while the atmospheric life of HFC32 is only five years.
Toxicity Ideally, refrigerants used in a refrigeration system should be non-toxic. However, all fluids other than air can be called as toxic as they will cause suffocation when their concentration is large enough. Thus toxicity is a relative term, which becomes meaningful only when the degree of concentration and time of exposure required to produce harmful effects are specified.
Toxicity • Some fluids are toxic even in small concentrations. Some fluids are mildly toxic, i.e., they are dangerous only when the concentration is large and duration of exposure is long. Some refrigerants such as CFCs and HCFCs are non-toxic when mixed with air in normal condition. However, when they come in contact with an open flame or an electrical heating element, they decompose forming highly toxic elements (e.g. phosgene-COCl2).
Toxicity • In general the degree of hazard depends on: • Amount of refrigerant used vs total space • Type of occupancy • Presence of open flames • Odor of refrigerant, and • Maintenance condition • Thus from toxicity point-of-view, the usefulness of a particular refrigerant depends on the specific application.
Flammability • The refrigerants should preferably be non-flammable and non-explosive. For flammable refrigerants special precautions should be taken to avoid accidents.
Chemical stability & Compatibility • The refrigerants should be chemically stable as long as they are inside the refrigeration system. • Compatibility with common materials of construction (both metals and non-metals)
Miscibility with lubricating oils • Oil separators have to be used if the refrigerant is not miscible with lubricating oil (e.g. ammonia). Refrigerants that are completely miscible with oils are easier to handle (e.g. R12). However, for refrigerants with limited solubility (e.g. R 22) special precautions should be taken while designing the system to ensure oil return to the compressor
Dielectric strength • This is an important property for systems using hermetic compressors. For these systems the refrigerants should have as high a dielectric strength as possible
Ease of leak detection • In the event of leakage of refrigerant from the system, it should be easy to detect the leaks.
Economic properties • The refrigerant used should preferably be inexpensive and easily available.
Halocarbon Refrigerants • Methane (CH4)Based will have a two-digit number ex:R22 First figure shows no. of Hydrogen atoms plus 1 Second figure shows no. of fluorine Atoms R22=CHClF2 Chlorofluoromethane. The total number of Hydrogen and replacement atoms should be 4; R11; R12; R13; R23 etc. • Ethane (C2H6) Based will have a three-digit number ex: R113. First figure shows no. of Carbon atoms minus 1; Second figure shows no. of Hydrogen atoms plus 1; Third figure shows no. of fluorine Atoms R113 =CCl2FCClF2 Tricholorofluoroethane; R114; R115 etc.
R134a=C2H2F4 Tetrafluoro ethane The letter a signifies isomer ( having same chemical composition but different atomic arrangement) • Refrigerants starting with 4 and 5 indicate blended refrigerants
The most important members of the group have been – • Chlorofluorocarbons (CFC) • Hydrochlorofluorocarbon (HCFC) • Hydrofluorocarbon(HFC)
Chlorofluorocarbons (CFC) • CCl2F2 - Dichlorodifluoromethane (Freon 12 or R12) • CCl3F -Trichlorofluoromethane (Freon 11 or R11) • C2Cl2F4 - Dichlorotetrafluoroethane (Freon 114 or R114) • C2Cl3F3 - Trichlorotrifluoroethane (Freon 113 or R113)
Hydrochlorofluorocarbon (HCFC) • CHClF2 – monochlorodifluoromethane (Freon 22 or R22) • R123 – C2HCl2F3 • R124 - C2HClF4
Hydrofluorocarbon(HFC) • C2H2F4 -R134a • C2H4F2 - R152a • C2HF5 - R125 • CH2F2 - R32 • C2H3F3 - R143a
Inorganic refrigerants • These are designated by number 7 followed by the molecular weight of the refrigerant (rounded-off).
Mixtures • Azeotropic mixtures (containing two gases with same boiling point ) are designated by 500 series, where as zeotropic (containing two or more gases not having same boiling point ) refrigerants (e.g. non-azeotropic mixtures) are designated by 400 series.
Depletion of stratospheric ozone layer • The depletion of stratospheric ozone layer was attributed to chlorine and bromine containing chemicals such as CFCs, HCFCs. If released to atmosphere, they are broken down by photolysis to release chlorine atoms, which catalytically destroy ozone, the stratospheric gas which acts as a filter to ultra violet (UV) light from the sun.
Effect of UV light • Scientists predict that increased UV light on earth as a result of ozone depletion will, amongst other possible consequences, cause skin cancer, interfere with immune systems and harm aquatic systems and crops. Strong pressure was exerted to phase out CFCs and HCFCs, resulted in the Montreal Protocol being adopted in 1987.
Regulation 12(2) of Annex VI of MARPOL 73/78 • New installations which contain ozone-depleting substances shall be prohibited on all ships, except that new installations containing hydrochloroflourocarbons (HCFCs) are permitted till 1 January 2020.
Alternate refrigerants • They can be classified into two broad groups: • Non-ODS, synthetic refrigerants based on Hydro-Fluoro-Carbons (HFCs) and their blends • Natural refrigerants including ammonia, carbon dioxide, hydrocarbons and their blends