Acid-Base Titrations
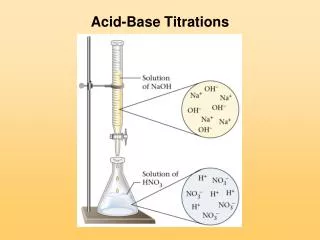
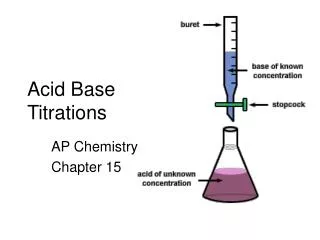
Acid-Base Titrations. Section 17.3. Introduction. Definition: In an acid-base titration , a solution containing a known concentration of a base is slowly added to an acid. An indicator is used to signal the equivalence point of the titration.

Acid-Base Titrations
E N D
Presentation Transcript
Acid-Base Titrations Section 17.3
Introduction • Definition: • In an acid-base titration, a solution containing a known concentration of a base is slowly added to an acid. • An indicator is used to signal the equivalence point of the titration. • This is the point at which stoichiometrically equivalent amounts of acid and base have been mixed. • A pH meter can also be used to find the equivalence point.
Introduction • The typical titration apparatus includes: • a buretto hold the titrant • a beaker to hold the analyte • a pH meter to measure the pH
Introduction • In this section, we will be looking at a series of titrations in detail to understand why acids and behave the way they do. • Strong acid-strong base titration • Weak acid-strong base titration • Polyprotic acid-strong base titration
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape.
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape. We divide the curve into four regions: 1. Initial pH 2. Initial pH to eq. point 3. Equivalence point 4. After eq. point
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape. We divide the curve into four regions: 1. Initial pH The pH of the solution is determined by the concentration of the strong acid.
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape. We divide the curve into four regions: 2. Initial pH to eq. point As base is added, pH increases slowly and then rapidly. The pH is determined by the concentration of the acid that is not neutralized.
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape. We divide the curve into four regions: 3. Equivalence point At the equivalence point, [OH−] = [H+]. The pH = 7.00.
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape. We divide the curve into four regions: 4. After eq. point As more base is added, pH increases rapidly and then slowly. pH is determined by the concentration of the excess base.
Strong Acid-Strong Base Titrations • The titration curve of a strong acid-strong base titration has the following shape. Let’s see how this works in practice.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL • 51.0 mL
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL This is between the initial point and the equivalence point.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL This is between the initial point and the equivalence point. pH is determined by the amount of acid that has not been neutralized.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL Therefore, we need to determine the number of mols of acid remaining, nacid, and the total volume, Vtotal, of the solution. (Remember, adding the NaOH increases the total volume.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= (0.100 M)(0.0500 L)
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= Mbase,added× Vbase,added
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= (0.100 M)(0.0490 L)
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= 4.90 × 10−3mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= 4.90 × 10−3mol nacid,remaining= nacid,i− nbase,added
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= 4.90 × 10−3mol nacid,remaining= (5.00 − 4.90) × 10−3mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= 4.90 × 10−3mol nacid,remaining= 0.10 × 10−3mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,i= Macid,i× Vacid,i= 5.00 × 10−3mol nbase,added= 4.90 × 10−3mol nacid,remaining= 1.0 × 10−4mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = Vacid + Vbase
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = Vacid + Vbase = 0.0500 L + 0.0490 L
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = Vacid + Vbase = 0.0990 L
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L [H+] = nacid,remaining/Vtotal
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L [H+] = (1.0 × 10−4mol)/(0.0990 L)
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L [H+] = 1.0 × 10−3 M
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L [H+] = 1.0 × 10−3 M pH = −log[H+]
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L [H+] = 1.0 × 10−3 M pH = −log(1.0 × 10−3)
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL nacid,remaining= 1.0 × 10−4mol Vtotal = 0.0990 L [H+] = 1.0 × 10−3 M pH = 3.00
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL pH = 3.00
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 49.0 mL • 51.0 mL
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL This is beyond the equivalence point.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL This is beyond the equivalence point. All of the strong acid has been used up.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL This is beyond the equivalence point. All of the strong acid has been used up. The pH is determined by the excess base that has been added.
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL We already determined nacid,i = 5.00 × 10−3mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL nacid,i = 5.00 × 10−3mol
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL nacid,i = 5.00 × 10−3mol nbase,added= Mbase,added× Vbase,added
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL nacid,i = 5.00 × 10−3mol nbase,added= (0.100 M)(0.0510 L)
Sample Exercise 17.6 (page 731) Calculate the pH when the following quantities of 0.100 M NaOH solution have been added to 50.0 mL of 0.100 M HCl solution. • 51.0 mL nacid,i = 5.00 × 10−3mol nbase,added= 5.10 × 10−3mol