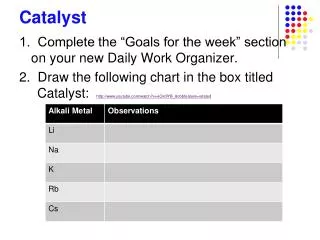
Catalyst
Catalyst. What is catalyst function?. Catalyst function is providing an alternative reaction pathway with lower activation energy so that more reaction particles possess necessary energy to react. Modes of action of homogenous catalysts.

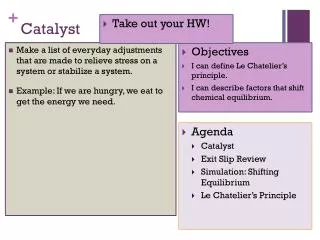
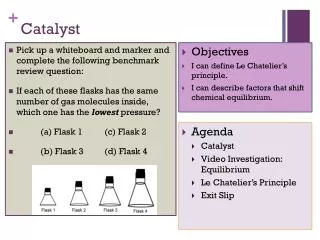
Catalyst
E N D
Presentation Transcript
What is catalyst function? • Catalyst function is providing an alternative reaction pathway with lower activation energy so that more reaction particles possess necessary energy to react
Modes of action of homogenous catalysts • Homogeneous catalysis is catalysis process where reactants and catalysts are in same state (liquid, gas, solid) • Homogeneous catalysts can form intermediate compounds during the reaction..
Modes of action of heterogeneous catalysts • Heterogeneous catalysts are in different phase (state) from the reactants • Heterogeneous catalysts function by absorbing reactant molecules onto the surface of the catalysts.
Advantages: Homogeneous- all the catalyst is exposed to the reaction Heterogeneous Easily removed from product by filtration Disadvantages Homogeneous--- hard to reuse Heterogeneous— only effective on the surface of product Outline disadvantages and advantages of homogeneous and heterogeneous catalysis
Selectivity – will be catalyst produce only the desired product? Efficiency - will the catalyst cause a considerable increase in the rate? - will it continue to work well under severe conditions, such as those experienced by catalytic converters in cars, as well as mild conditions Environmental impact – will it be easy to dispose of the catalyst without causing harm to the environment? Potential for poisoning – catalysts rely on reactant occupying the active site reversibly. Cost – industry is profit –based. Discuss the factors in choosing a catalyst for a process