The SDS-PAGE
250 likes | 820 Views
ABE Workshop 2006. The SDS-PAGE. Dongping Lu. Western blotting. Trouble shooting. 1. We should have a parameter: Total protein /plant tissue 2. Use higher concentration of BSA Standard, like BSA 10mg/ml. Absorption value. BSA. Today’s work. Run the SDS-PAGE gel:

The SDS-PAGE
E N D
Presentation Transcript
ABE Workshop 2006 The SDS-PAGE Dongping Lu Dongping Lu ABE Workshop June 21, 2006
Trouble shooting 1. We should have a parameter: Total protein /plant tissue 2. Use higher concentration of BSA Standard, like BSA 10mg/ml Absorption value BSA
Today’s work • Run the SDS-PAGE gel: • Transfer the protein from gel to nitrocellulose membrane • Stain the gel with Coomassie Blue group 1 & 4: wt and pdi2 mutant group 2 & 3: wt and gfp-2sc
PAGE • Gels are cast by polymerizing a solution of acrylamide monomers into polyacrylamide chains • Gel pore size can be varied by adjusting the concentrations of polyacrylamide • Smaller proteins migrate faster than larger proteins through the gel
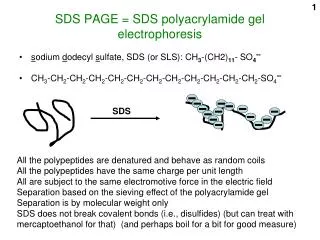
1. SDS disrupts some of the noncovalent interactions that stabilize protein quaternary and tertiary structures, facilitates denaturation. 2. SDS also has a negative electrical charge and binds to proteins in a constant mass ratio of 1.4 : 1, so that the total amount of detergent bound is directly proportional to the molecular weight of the protein. 3. The ‘coating’ of negatively charged SDS overwhelms the inherent charges of protein molecules and gives them a uniform charge to mass ratio. 4. This allows proteins to be separated on the basis of their relative sizes, SDS
SDS • all polypeptide chains are then forced into extended conformations • SDS treatment eliminates the effect of differences in shape • individual polypeptide chains migrate as a negatively charged SDS-protein complex through the porous polyacrylamide gel • speed of migration is proportional to the size of the proteins • smaller polypeptides running faster than larger polypeptides
How about covalent link? DTT/Me SH S-S HS
Noncovalent covalent
Heating the sample • Heating your samples at 99ºC completed denaturation of the protein molecules, ensuring that they were in completely linear form. • This allowed SDS to bind all regions of each protein equally.
Protein loading buffer • Protein gel loadingbuffer contains Tris buffer to maintain constant pH • glycerol to increase sample density, • the strong ionic detergent SDS (sodium dodecylsulfate), • β-mercaptoethanol, a reducing agent. . Beta-mercaptoethanol eliminates disulfide bonds in proteins by reducing them (adding hydrogen atoms). • Heating
Stacking gel • To obtain optimal resolution of proteins, a “stacking” gel is poured over the top of the “resolving” gel. • The stacking gel lower concentration of acrylamide (larger pore size), lower pH different ionic content • This allows the proteins in a lane to be concentrated into a tight band before entering the running or resolving gel • produces a gel with tighter or better separated protein bands
Transfer the protein from the gel to the membrane • Transfer of the proteins fractionated by SDS-PAGE to a solid support membrane (Western blotting) can be accomplished by electroblotting
Transfer • In this procedure, a sandwich of gel and solid support membrane (Nitrocellulose or PVDF) is compressed in a cassette and immersed in buffer between two parallel electrodes. • A current is passed at right angles to the gel, which causes the separated proteins to electrophorese out of the gel and onto the solid support membrane
Gel staining • Once proteins have been fractionated by electrophoresis, to make them visible, staining with a material that will bind to proteins but not polyacrylamide. • the most common one: staining with Coomassie Blue. • This is a dye that binds most proteins uniformly based on interactions with the carbon-nitrogen backbone. • The dye is dissolved in a solution that contains both methanoland acetic acid
Gel drying • SDS-PAGE gels between two moistened sheets of Gel Drying Film (from Promega) on the bench. • Clamp the Gel Drying Frame • Dry over night • It is important to remove all the air bubbles from between the two sheets of gel drying films. Air bubbles may cause the gel to crack during drying