VSEPR
VSEPR . Valence Shell Electron Pair Repulsion. Electron pairs orient themselves so as to make the angles between themselves as large as possible Repulsion follows Coulomb’s law This includes bonded as well as non-bonded pairs. Molecular Geometry.

VSEPR
E N D
Presentation Transcript
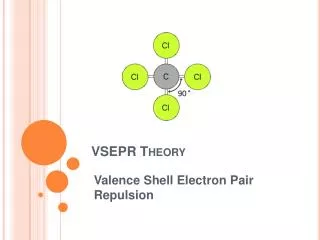
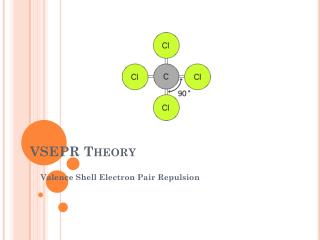
Valence Shell Electron Pair Repulsion • Electron pairs orient themselves so as to make the angles between themselves as large as possible • Repulsion follows Coulomb’s law • This includes bonded as well as non-bonded pairs
Molecular Geometry • Non-bonded pairs are not seen on the molecule but they do affect it • In determining molecular geometries, multiple bonds are considered like single bonds • Six types of molecular geometries
Electron Pair Geometry • Geometry if unshared pair thought of as a bond
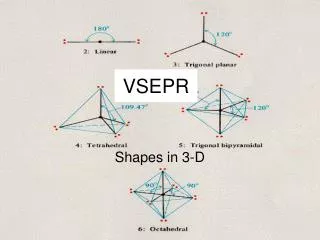
Linear Molecular and Electron-Pair Geometry • Two bonded pair and no non-bonded pairs on central atom • 180°
TrigonalPlaner Molecular and Electron-Pair Geometry • Three bonded pair and no non-bonded pairs • 120° between bonds
Bent Molecular and Trigonal Planer Electron-Pair Geometry • Two bonded pair and one non-bonded pair • 120° between bonds
Tetrahedral Molecular and Electron-Pair Geometry • Four bonded pairs • 109.5° between bonds
TrigonalPyramidal Molecular and Tetrahedral Electron-Pair Geometry • Three bonded and one non-bonded • 109.5° between bonds
Bent Molecular and Tetrahedral Electron-Pair Geometry • Two bonded and two non-bonded • 104.5° between bonds
Non-Bonding Pairs • Exert greater repulsive forces on adjacent pairs than bonded electrons do • Example – All of these have tetrahedral electron pair geometries • CH4 109.5° • NH3 107° • H2O 104.5°