4. Radioactive Decay
4. Radioactive Decay. radioactive transmutation and decay are synonymous expressions 4 main series 4n 232 Thorium 4n + 2 238 Uranium-Radium 4n + 3 235 Actinium 4n + 1 237 Neptunium. 4.1 Decay Series. 4.2a Law and Energy of Radioactive Decay.

4. Radioactive Decay
E N D
Presentation Transcript
4. Radioactive Decay • radioactive transmutation and decay are synonymous expressions • 4 main series 4n 232Thorium 4n + 2 238Uranium-Radium 4n + 3 235Actinium 4n + 1 237Neptunium 4.1 Decay Series
4.2a Law and Energy of Radioactive Decay • radioactive decay law follows Poisson statistics behaves as where: N is the number of atoms of a certain radionuclide; -dN/dt is the disintegration rate; and is the disintegration constant in sec-1
4.2a Law and Energy of Radioactive Decay • law of radioactive decay describes the kinetics of a reaction Where A is the mother radionuclide; B is the daughter nuclide; X is the emitted particle; and E is the energy set free by the decay process (also known as Q-value)
4.2a Law and Energy of Radioactive Decay • radioactive decay only possible when E > 0 which can be calculated as • however decay may only arise if nuclide A surmounts an energy barrier with a threshold ES or through quantum mechanical tunneling
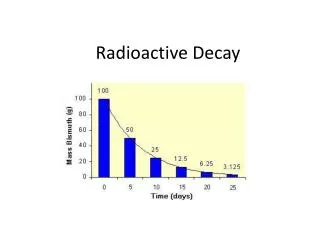
4.2b Kinetics of Radioactivity Half-Life • the time for any given radioisotope to decrease to 1/2 of its original quantity • range from a few microseconds to billions of years
4.2b Kinetics of Radioactivity t1/2 = 5 years
4.2b Kinetics of Radioactivity • each isotope has its own distinct half-life (t1/2) and in almost all cases no operation, physical or chemical, can alter the transformation rate 1st half-life 50% decay 2nd half-life 75% decay 3rd half-life 87.5% decay 4th half-life 93.75% decay 5th half-life 96.87% decay 6th half-life 98.44% decay 7th half-life 99.22% decay
4.2c Probability of Disintegration • number of nuclei dN in a time interval dt will be proportional to that time interval and to the number of nuclei N that are present; or at any time t there are N nuclei dN = - Ndt • where is the proportionality constant and the -ve sign is introduced because N decreases
4.2c Probability of Disintegration • at t = 0: N = N0 therefore lnN0 = C • the fraction of any radioisotope remaining after n half-lives is given by
4.2c Probability of Disintegration • where No is the original quantity and N is the quantity after n half lives
4.2c Probability of Disintegration • if the time t is small compared with the half-life of the radionuclide ( t<<t1/2)then we can approximate
4.2c Probability of Disintegration Average Life of an Isotope • it is equally important to know the average life of an isotope
4.2c Probability of Disintegration Decay Constant Problems • what is the constant 52V which has a t1/2 = 3.74 min.?
4.2c Probability of Disintegration • what is the constant for 51Cr which has a t1/2 = 27.7 days? • what is the constant for 226Ra which has t1/2 = 1622 yrs
4.2c Probability of Disintegration Decay Problem • what % of a given amount of 226Ra will decay during a period of 1000 years? 1/2 life of 226Ra = 1622 yr
4.2c Probability of Disintegration • therefore the percentage transformed during the 1000 year period is: 100% - 64.5% = 35.5%
4.2d Activity • Curie (Ci), originally defined as the activity of 1 gm of Ra in which 3.7 1010 atoms are transformed per sec • in S.I. units activity is measured in Becquerel (Bq), where 1 Bq = 1 tps -> the quantity of radioactive material in which one atom is transformed per sec
4.2d Activity activity of a radionuclide is given by its disintegration rate
4.2d Activity • equal weights of radioisotopes do not give equivalent amounts of radioactivity • 238U and its daughter 234Th have about the same no. of atoms per gm. However their half- lives are greatly different • 238U = 4.5 109 yr; 234Th = 24.1 days • therefore, 234Th is transforming 6.8 1010 faster than 238U
60Co , 0.314 MeV , 1.1173 MeV 60Ni , 1.332 MeV 4.2d Activity 1 Bq with 3 emissions
42K , 2.04 MeV 18% , 1.53 MeV 42Ca 4.2d Activity 1 Bq with 1.18 emissions
4.2d Activity 1 kilobecquerel (kBq) = 103 Bq 1 megabecquerel (MBq) = 106 Bq 1 gigabecquerel (GBq) = 109 Bq 1 terabecquerel (TBq) = 1012 Bq 1 millicurie (mCi) = 10-3 Ci 1 microcurie (μCi = 10-6 Ci 1 nanocurie (nCi) = 10-9 Ci 1 picocurie (pCi) = 10-12 Ci 1 femtocurie (fCi) = 10-15 Ci 1 Ci = 3.7 1010 Bq
4.2d Activity • since activity A is proportional to N, the number of atoms, we get A = A0e-t • the mass m of radioactive atoms can be calculated from their number N; activity A; M mass of nuclide; and Nav Avogadro’s number • ( 6.02 X 1023)
4.2d Activity Problem ● how much time is required for 5 mg of 22Na (t1/2 = 2.60 y) to reduce to 1 mg? ● since the mass of a sample willbe proportional to the no. of atoms in the sample get
4.2d Activity Specific Activity • the relationship between mass of the material and activity or AS (SA) = no. of Bq's/unit mass or volume
4.2d Activity • SA can also be represented in combined mathematical known terms
4.2d Activity • SA may also be derived by using the fact that there are 3.7 1010 tps in 1 gm of 226Ra
4.2d Activity Problem • calculate the specific activity of 14C (t1/2 = 5730 yrs)
4.2d Activity Problem • potassium (atomic weight = 39.102 AMU) contains: • 93.10 atom % 39K, having atomic mass 38.96371 AMU • 0.0118 atom % 40K, which has a mass of 40.0 AMU and is radioactive with: t1/2 = 1.3 109 yr • 6.88 atom % 41K having a mass of 40.96184 AMU
4.2d Activity • estimate the specific activity of naturally occurring potassium • specific activity refers to the activity of 1 g material • 1 g of naturally occurring potassium contains: 1.18 10-4 g 40K plus non-radioactive isotopes
4.2d Activity Problem • prior to use of nuclear weapons, the SA of 14C in soluble ocean carbonates was found to be 16 dis/min ·g carbon • amount of carbon in these carbonates has been estimated as 4.5 1016 kg • how many MCi of 14C did the ocean carbonates contain?
4.2d Activity Problem • a mixture of 239Pu and 240Pu has a specific activity of 6.0 109 dps • the half-lives of the isotopes are 2.44 104 and 6.58 103y, respectively • calculate the isotopic composition
4.2d Activity • for 239Pu • for 240Pu
4.2d Activity • number of seconds in a year is • for 239Pu: A = 2.27 109/s g • for 240Pu: A = 8.37 109/s g • let the fraction of 239Pu = x; then the fraction 240Pu = 1 - x
4.2d Activity (2.27 109)x+(1 – x)(8.37 109) = 6.0 109 (8.37 109) – (6.10 109) x = 6.0 109 2.37 109 = (6.1 109) x x = 0.39 = 39% 239Pu
4.2d Activity Problem • if 3 10-9 kg of radioactive 200Au has an activity of 58.9 Ci, what is its half-life? • no. of atoms in 3 10-9 kg of 200Au is
4.2d Activity • decay constant is found from A = N • finally
4.3 Radioactive Equilibria • net production of nuclide 2 is given by decay rate of nuclide 1 less the decay rate of nuclide 2
4.3 Radioactive Equilibria • given that: • solution of first order differential equation
4.3 Radioactive Equilibria • if nuclide 1 and 2 are separated at t = 0; then nuclide 2 is not produced and
4.3 Radioactive Equilibria • after substitution for λ: • the exponent term can be written to show the influence the ratio of
4.4 Secular Equilibrium • in secular equilibrium t1/2 (1)>> t1/2 (2) so • reduces