Health security
440 likes | 467 Views
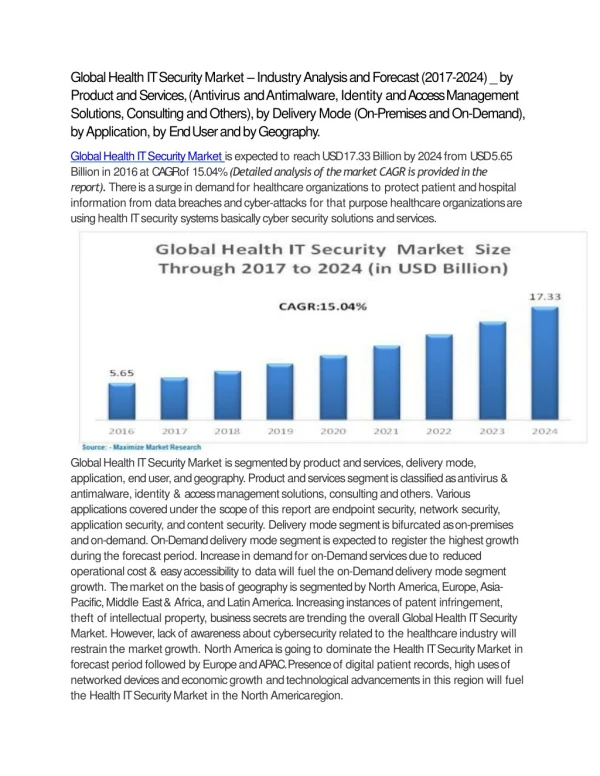
Health security. Haraldur Briem Chief Epidemiologist. Global public health security, 1980-2008. 1981. Influenza pandemic 2009. The attack on the World Trade Center 11 September 2001. The aftermath of 11 September 2001.

Health security
E N D
Presentation Transcript
Health security Haraldur Briem Chief Epidemiologist
Global public health security, 1980-2008 1981 Influenza pandemic 2009
The aftermath of 11 September 2001 Mediastinal widening and pleural effusionon Chest X-Ray in inhalational anthrax Mediastinal widening with inhalational anthrax (JAMA 1999:281:1735-1745)
Varicellae Smallpox Smallpox – will we recognize it ?
2 júní 1707 The Great smallpox epidemic in Iceland 1707-1708 Desember 1707-Janúar 1708? Ágúst-desember 1707 Ágúst-desember 1707 Ágúst 1708 Maí 1708 Ágúst-desember 1707 Janúar 1708 ? Alþingi 8.-26. júlí 1707 Desember 1707 Júlí 1707 Eyrarbakki
2 júní 1707 ? Desember 1707-Janúar 1708? Ágúst-desember 1707 Ágúst-desember 1707 Ágúst 1708 Maí 1708 Ágúst-desember 1707 Janúar 1708 ? Alþingi 8.-26. júlí 1707 Desember 1707 Júlí 1707 Eyrarbakki
Mortality ratio (%) due to smallpox in Álftanes County from 1 September til 27 November 1707. Population 576 of whom 172 died (30%). Vertical bars indicate 95% confidence intervals 60 50 40 % 30 20 10 0 0-4 5-9 10-19 20-29 30-39 40-49 50-59 >60 Age groups
Avian influenza H5N1 Ducks and Other birds
The Spanish flu in IcelandOctober - December 1918 Reykjavík: Attack rate: 63 % Mortality rate: 17 per 1000 population Case fatality rate: 2,8%
Radiological Weapons • On 1 November 2006, former lieutenant colonel of the Russian Federal Security Service Alexander Litvinenko suddenly fell ill and was hospitalised. He died three weeks later, becoming the first known victim of lethal polonium-210 induced acute radiation syndrome
The Scope of the IHR • to prevent, protect against, control and provide a public health response to the international spread of disease in ways that are commensurate with and restricted to public health risks, and which avoid unnecessary interference with international traffic and trade. • global health security: • epidemic alert and response, • global public health response to natural occurrence, accidental release or deliberate use of biological and chemical agents or radio nuclear material that affect health, to the need to ensure global public health
What is new? • From three diseases to all public health threats • From preset measures to adapted response • From control of borders to, also, containment at source
Act on Health Security and Communicable Diseases, nr. 19/1997, revised 2007 The scope: • Threats to public health • Epidemic diseases. • Serious diseases caused by infectious agents, microbes, parasites, toxic agents and radio-nuclear substances. • Unusual or unexpected events that may be of international concern.
National IHR Focal Point • National IHR Focal Point” means the national centre, designated by each State Party, which shall be accessible at all times for communications with WHO IHR Contact Points under these Regulations • “WHO IHR Contact Point” means the unit within WHO which shall be accessible at all times for communications with the National IHR Focal Point.
Some milestones • March 2005: The Icelandic government appointed a committee composed of Ministerial Directors General to review the situation in Iceland with regard to a potential influenza pandemic • October 2005: The Icelandic government approved the proposals of responses and measures with regard to a influenza pandemic. • February 2006: The Government decided to entrust to the National Commissioner of the Icelandic Police and the Chief Epidemiologist the establishment of a steering group to define the assignments for civil protection operations with regard to responses to the threat of pandemic influenza.
Preparedness and Response to Pandemic Influenza in Iceland The Government The Ministry of Justice The Ministry of Health Chief Epidemiologist Early Warning and Response National Commissioner of Police Department of Civil Defense • The Health Care System. • Specific resources. • Public measures against communicable diseases: • Immunizations, • Isolation, • Quarantine measures of individuals, geographic regions, border closure, closure of schools and mass gatherings Essential infrastructure of the society General resources. Assistance for the public measures against communicable diseases
Early Warning and Response Surveillance Hospitals Health Officials Veterinarians Radiation Agency Toxic substances Occupational Health Communication Travel Industry Police Rumor Media WHO ECDC r National Focal Point Minister/ Chief Epidemiologist SSUS Assesment Health Officials Hospitals Veterinarians Radiation Toxins Activities Communication Travel Industry Police. Response ECDC WHO
Regions for Containment of Communicable Diseases Pandemic influenza 2009in Iceland Centre for Health Security and Communicable Disease Control Directorate of Health
Preparedness plan activated Alert given in end of April 2009 Entry screening? Severity assessment in May 2009 Drug and equipment (PPE) distribution WHO declares pandemic influenza in June 2009 July influenza starts in Iceland
Heildarfjöldi barna í Grunnskóla Siglufjarðar var 168 á þessu tímabili. Weekly absence of students in an elementary school in Northern Iceland from 28 September to 30 October 2009 Per cent of 168 students Prósentuhlutfall af 168 nemendum W 40 W 41 W 43 W 45 W46
Influenza A(H1N1)v 2009 • 17-20% of the population infected • ~200 patients hospitalized (3,6/1000) • At least two deaths due to verified influenza • 40% of the population vaccinated • 10.014 antiviral treatment doses given • Preventive measures may have protected • 10.000-30.000 people from disease • 36-100 hospital admissions and • at least one death
The Climate Problem • Present global temperatures are in a warming phase that began 200 to 300 years ago. • Climate models suggest that human activities may have exacerbated this phase by raising the atmospheric concentration of carbon dioxide and other greenhouse gases. • A decline in temperatures from the 1940s to the late 1970s gave rise to warnings that industrial pollutants were causing global cooling.
Climate Change and Health • Climate change may lead to diseases and death by causing: • Heat waves • Flooding • Draft • Rainfall • Increased ozone on the surface of the earth, pollen in the air and other air pollution • Increased incidence of infectious diseases
Climate Change and Health • Health consequences • Injuries • Famine • Heart diseases • Respiratory diseases • Infectious diseases • Animal- and plant diseases • Categories of consequences • Non infectious diseases • Infectious diseases
WHO estimate on death rate due to climate change in 2000 Death rate/million 0-2 2-4 4-70 70-120 No information
Determinants for Emerging Infectious Diseases • Transmission routes and virulence • Changes in the environment (such as hygiene, sanitation, climate change) • Changes in the host, human behavior, age at infection, population density, migration • Old infectious microorganisms with new virulent properties such as antimicrobial resistance • New microorganisms • Zoonotic diseases (wildlife – non wildlife)
Emerging Infectious Diseases • Newly evolved strains of pathogens (for example, multi-drug-resistant tuberculosis and chloroquine-resistant malaria) • Pathogens that have recently entered human populations for the first time (for example, HIV-1, severe acute respiratory syndrome (SARS) coronavirus) • Pathogens that have probably been present in humans historically, but which have recently increased in incidence (for example, Lyme disease) Jones KE et al. Global trends in emerging infectious diseases NATURE 2008;451:990-993
Global trends in emerging infectious diseases (EID) • 60.3% of EID events are dominated by zoonoses • Driven largely by socio-economic, environmental and ecological factors • EID events have risen significantly over time (1940-2004) • The majority of zoonotic EID events (71.8%) originate in wildlife (for example, coronavirus causing severe acute respiratory syndrome, Ebola virus), and are increasing significantly over time. Jones KE et al. Global trends in emerging infectious diseases NATURE 2008;451:990-993
The map is derived for EID events caused by all pathogen types. Circles represent one degree grid cells, and the area of the circle is proportional to the number of events in the cell.
a, pathogen type b, transmission type c, drug resistance d, transmission mode
The climate–dengue debate • Dengue fever is the most prevalent mosquito-borne viral disease in the world, causing an estimated 50 million infections and 25,000 deaths annually, with at least 2.5 billion persons at risk for transmission • Dengue fever transmission on the US side of the Texas–Mexico border have been rare—only 64 cases were reported during 1980–1999, compared with 62,514 cases on the Mexican side of the border • In the debate over the potential for expansion of dengue and malaria with climate change, the border region has been cited as evidence that mosquito-borne diseases are largely determined by public health capacity and socioeconomic factors, and specifically that US affluence and lifestyle limit transmission of the disease Marie Brunkard, JM et al. Dengue Fever Seroprevalence and Risk Factors, Texas–Mexico Border, 2004. Emerg Infect Dis 2007: 13:1478-1483.
Map of Brownsville, Texas, and Matamoros, Mexico, contiguous cities on the US–Mexico border.
Dengue in Brownsville/Matamoros Results • Recent dengue infection in residents • of Brownsville 2% (95% CI 0.5%–3.5%) • of Matamoros 7.3% (95% CI 4.3%–10.3%) • Past infection in residents • of Brownsville 40% (95% CI 34%–45%) of Matamoros 78% (95% CI 74%–83%) Marie Brunkard, JM et al. Dengue Fever Seroprevalence and Risk Factors, Texas–Mexico Border, 2004. Emerg Infect Dis 2007: 13:1478-1483.
Dengue in Brownsville/Matamoros Results • For recent infection, only weekly family income <$100 was a significant predictor (adjusted odds ratio 3.2, 95% CI 1.3–8.0). • Risk factors that predicted past dengue infection were presence of • larval habitat • absence of air-conditioning and street drainage • weekly family income <$100. Marie Brunkard, JM et al. Dengue Fever Seroprevalence and Risk Factors, Texas–Mexico Border, 2004. Emerg Infect Dis 2007: 13:1478-1483.
Richter scale representing a snapshot in time (2003) of natural “weapons” of mass destruction Twenty years ago, HIV was three logs further down the scale, whereas polio was three logs higher. Among the chronic and re-emerging infections, malaria and tuberculosis are near the top Weiss, RA & McMichael, AJ. Social and environmental risk factors in the emergence of infectious diseases Nature Medicine 2004;10:S70-76
Conclusion • The greatest health security risk for Iceland is from mother nature be it epidemics or other natural disasters • There is a security risk due to intentional or accidental releases of biological, chemical or radio-nuclear agents • There is an insignificant risk of military attack on Iceland by a foreign state military power • It is a mistake to separate military defense from civil defense – it is the 19th and 20th centuries way of thinking – ignoring the realities of the 21st century